Advertisements
Advertisements
Question
Give an example for each of the following statement.
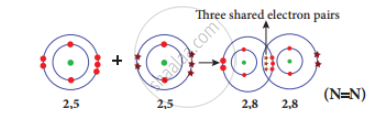
A compound in which three covalent bonds are formed.
Advertisements
Solution
Formation of nitrogen molecule (N2): Nitrogen molecule (N2) is formed by three covalent bonds where three pairs of electrons are shared between the two to achieve a stable configuration.
APPEARS IN
RELATED QUESTIONS
Explain the nature of the covalent bond using the bond formation in CH3Cl.
Give one example of a molecule containing a double covalent bond
Describe the structure of diamond. Draw a simple diagram to show the arrangement of carbon atoms in diamond.
For each compound mentioned above give the formulae of ions formed in aqueous solution.
Give two example in following case:
Liquid non polar compounds
Draw electron - dot structure and structural formula of methane.
What do you understand by lone pair and shared pair?
Fill in the blank with correct word from the bracket.
Melting and boiling points of covalent compounds are generally ______ (low, high).
Fill in the blank from the choice given in bracket.
The compound that does not have a lone pair of electrons is ___________. (Water, Ammonia, carbon tetrachloride)
Statement (A): Covalent compounds are bad conductors of electricity.
Reason (B): Covalent compounds contain charged particles (ions)
