Advertisements
Advertisements
प्रश्न
Give an example for each of the following statement.
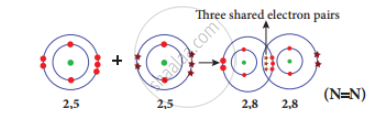
A compound in which three covalent bonds are formed.
Advertisements
उत्तर
Formation of nitrogen molecule (N2): Nitrogen molecule (N2) is formed by three covalent bonds where three pairs of electrons are shared between the two to achieve a stable configuration.
APPEARS IN
संबंधित प्रश्न
An element L consists of molecules.
What type of bonding is present in the particles that make up L?
What is buckminsterfullerene?
Which of the following has a triple bond as well as single bonds?
(a) ethene
(b) methane
(c) ethyne
(d) nitrogen
State the type of bonding in the following molecule.
Ammonium ion
Give two example in following case:
Liquid non polar compounds
Explain the Structural isomerism term with example.
Draw the electron dot diagram and structure of magnesium chloride.
Name two compounds that are covalent when taken pure but produce ions when dissolved in water.
Name the following:
\[\ce{CH3 - CH2CH = CH2}\]
Show the covalent bond formation in nitrogen molecule.
