Advertisements
Advertisements
प्रश्न
Give an example for each of the following statement.
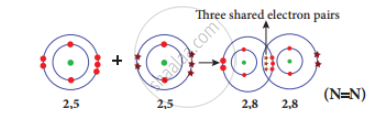
A compound in which three covalent bonds are formed.
Advertisements
उत्तर
Formation of nitrogen molecule (N2): Nitrogen molecule (N2) is formed by three covalent bonds where three pairs of electrons are shared between the two to achieve a stable configuration.
APPEARS IN
संबंधित प्रश्न
Explain the bonding in methane molecule using electron dot structure.
what substance is graphite made?
What are the conditions necessary for the formation of covalent molecules?
Explain the structure of Ammonium ion.
Explain the bonding in methane molecule using electron dot structure.
Draw an electron dot diagram to show the formation of the following compound.
Methane
Write an Explanation.
Alkane
Write scientific reason.
Benzene compounds are called aromatic compounds.
Write a short note.
Aromatic hydrocarbons
Non-polar covalent compounds are ______ conductors of heat and electricity.
