Advertisements
Advertisements
Questions
Explain the following, giving two examples:
Coordination number
Explain with two examples of the following:
Coordination number
Advertisements
Solution
It is the number of ligand donor atoms to which the metal is directly bonded. For example:
- Cu(NH3)4]2+: In this complex, the coordination number of copper (Cu2+) is 4 because there are four ammonia (NH3) molecules bonded to the metal ion.
- [Fe(CN)6]4−: Here, the coordination number of iron (Fe3+) is 6, as six cyanide (CN−) ions are coordinated to the iron ion.
APPEARS IN
RELATED QUESTIONS
IUPAC name of the following compound is
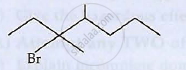
(a) 3 - Bromo- 3, 4- dimethylheptane
(b) 3, 4- dimethyl - 3- bromoheptane
(c) 5- Bromo- 4, 5- dimethylheptane
(d) 4, 5- dimethyl- 5- bromoheptane
Write the IUPAC name of the following complex:
[Co(NH3)5(CO3)]Cl
Complete the following reactions
NH3+3Cl2(excess) ---->
Write the IUPAC name of the following coordination compound:
[PtCl2(NH3)4][PtCl4]
The coordination number of Cr in [Cr(NH3)3(H2O)3]Cl3 is ___________.
In which of the following compounds, the Central metal atom/ion is in the lowest oxidation state?
Which of the following complexes formed by \[\ce{Cu^2+}\] ions is most stable?
A complex of the type \[\ce{[M(AA)2X2]^{n+}}\] is known to be optically active. What does this indicate about the structure of the complex? Give one example of such complex.
Match the compounds given in Column I with the oxidation state of cobalt present in it (given in Column II) and assign the correct code:
| Column I (Compound) | Column II (Oxidation state of Co) |
| A. \[\ce{[Co(NCS)(NH3)5](SO3)}\] | 1. + 4 |
| B. \[\ce{[Co(NH3)4 CL2]SO4}\] | 2. 0 |
| C. \[\ce{Na4[Co(S2O3)3]}\] | 3. + 1 |
| D. \[\ce{[Co2(CO)8]}\] | 4. + 2 |
| 5. + 3 |
Assertion: \[\ce{Cr(H2O)6]Cl2 and [Fe(H2O)6]Cl2}\] are reducing in nature.
Reason: Unpaired electrons are present in their d-orbitals.
What is the relationship between observed colour of the complex and the wavelength of light absorbed by the complex?
The oxidation state of Fe in the brown ring complex [F3(H2O)5NO]SO4 is
The most stable ion is ______.
How many ions are produced from the complex Co(NH3)6]Cl2 in solution?
The following ligand is:

The correct order of the spin-only magnetic moment of metal ions in the following low spin complexes,
\[\ce{[V(CN)6]^{4-}, [Fe(CN)6]^{4-}, [Ru(NH3)6]^{3+} and [Cr(NH3)6]^2}\] is:
What is meant by the chelate effect? Give an example.
Explain the following, giving two examples:
Coordination polyhedron
Give two examples of didentate ligands.
Give two examples of ambidentate ligands.
