Advertisements
Advertisements
Question
Draw the electron-dot structure of a hydrogen chloride molecule.
Advertisements
Solution
The electron-dot structure of a hydrogen chloride molecule is:
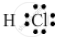
It is formed by sharing one electron by the hydrogen and chlorine atoms.
APPEARS IN
RELATED QUESTIONS
Fill in the blank in the following sentence:
The number of single covalent bonds in C2H2 molecule are ...........
What is the atomic number of carbon. Write its electronic configuration.
Explain why covalent compounds have generally high melting points?
How will you find out which of the water soluble compound A or B is ionic?
Describe the structure of graphite with the help of a labelled diagram.
Why are covalent compounds generally poor conductors of electricity?
The electronic configurations of three elements X, Y and Z are as follows:
| X | 2, 4 |
| Y | 2, 7 |
| Z | 2, 1 |
(a) Which two elements will combine to form an ionic compound?
(b) Which two elements will react to form a covalent compound?
Give reasons for your choice.
The pencil leads are made of mainly:
(a) lithium
(b) charcoal
(c) lead
(d) graphite
Draw an electron dot structure of the following molecule. (Without showing the circle) :
Ethene
Draw electron - dot structure and structural formula of methane.
Choose and write the correct option.
Which type of carbon-carbon bonds are present in Vanaspati ghee?
Give examples for the following:
Two liquid non polar compounds.
Explain the following:
Covalent compounds have low melting and boiling point.
Compare the compounds carbon tetrachloride and sodium chloride with regard to solubility in water and electrical conductivity.
An element L consists of molecules.
What type of bonding is present in the particles that make up L?
In the formation of electrovalent compounds, electrons are transferred from one element to another. How are electrons involved in the formation of a covalent compound?
Name two carbon compounds used in day-to-day life.
The number of electrons in the valence shell of a carbon atom is 4.
Name the following:
\[\ce{CH3 - CH2CH = CH2}\]
