Advertisements
Advertisements
प्रश्न
Draw the electron-dot structure of a hydrogen chloride molecule.
Advertisements
उत्तर
The electron-dot structure of a hydrogen chloride molecule is:
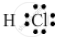
It is formed by sharing one electron by the hydrogen and chlorine atoms.
APPEARS IN
संबंधित प्रश्न
What are covalent compounds?
State the reason why carbon can neither form C4+ cations nor C4− anions but forms covalent compounds.
What would be the electron-dot structure of carbon dioxide which has the formula CO2?
What type of chemical bond is formed between carbon and bromine?
Give one example of a molecule containing a triple covalent bond.
What is the atomic number of carbon. Write its electronic configuration.
will CS2 conduct electricity or not?
give reason for your choice.
Give the formula of the compound that would be formed by the combination of the following pair of elements:
Al and Cl2
Which of the following cannot exhibit isomerism?
(a) C4H10
(b) C5H12
(c) C3H8
(d) C6H14
Explain the following:
polar covalent compounds electricity.
Draw electron - dot structure and structural formula of methane.
Explain the following:
Polar covalent compounds are good conductors of electricity.
In the formation of electrovalent compounds, electrons are transferred from one element to another. How are electrons involved in the formation of a covalent compound?
Draw an electron dot diagram to show the formation of the following compound.
Magnesium chloride [ H=1, C=6, Mg=12, Cl=17].
Electrons are getting added to an element Y:
which electrode will Y migrate to during the process of electrolysis?
Element A has 2 electrons in its M shell. Element B has atomic number 7.
If the compound formed between A and B is melted and an electric current is passed through the molten compound, the element A will be obtained at the ______ and B at the ______ of the electrolytic cell.
Two carbon atoms can always form one or two covalent bonds.
"Carbon prefers to share its valence electrons with other atoms of carbon or with atoms of other elements rather than gaining or losing the valence electrons in order to attain noble gas configuration." Give reasons to justify this statement.
Explain dipole (polar) molecule by taking hydrogen chloride as an example.
