Advertisements
Advertisements
प्रश्न
Draw the electron-dot structure of a hydrogen chloride molecule.
Advertisements
उत्तर
The electron-dot structure of a hydrogen chloride molecule is:
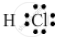
It is formed by sharing one electron by the hydrogen and chlorine atoms.
APPEARS IN
संबंधित प्रश्न
State the type of bonding in the following molecule.
Water
What type of bonds are present in CO2 molecule? Draw their electron-dot structures.
Give one example of a molecule containing a double covalent bond
Fill in the blank in the following sentence:
Two chlorine atoms combine to form a molecule. The bond between them is known as ..........
What type of bond exist in CCl4?
What is buckminsterfullerene?
How will you find out which of the water soluble compound A or B is ionic?
Define a coordinate bond.
What do you understand by lone pair and shared pair?
Explain the structure of Ammonium ion.
(a) Compound X consists of molecules.
Choose the letter corresponding to the correct answer from the choices (a), (b), (c) and (d) given below
In the liquid state, X will
Fill in the blank with correct word from the bracket.
Melting and boiling points of covalent compounds are generally ______ (low, high).
An element L consists of molecules.
Why L is heated with iron metal, it forms a compound FeL. What chemical term would you use to describe the change undergone by L?
State the type of bonding in the following molecule.
Water
Name gas with a rotten egg smell.
Fill in the blank and rewrite the completed statement:
Covalent compounds are generally soluble in _________ solvents.
The number of pi-bonds present in benzoic acid molecule are ______.
Which of the following statements are usually correct for carbon compounds? These
- are good conductors of electricity
- are poor conductors of electricity
- have strong forces of attraction between their molecules
- do not have strong forces of attraction between their molecules.
It is observed that covalent compounds are had conductors of electricity. Give reason.
In covalent compounds, the bond is formed due to ______ of electrons.
