Advertisements
Advertisements
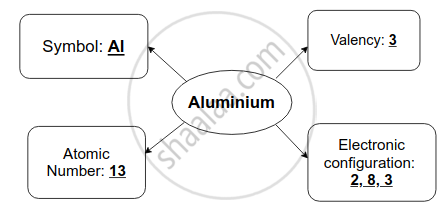
Question
Complete flow chart given below.

Advertisements
Solution
RELATED QUESTIONS
M is a metal above hydrogen in the activity series and its oxide has the formula M2O. This oxide when dissolved in water forms the corresponding hydroxide which is a good conductor of electricity. In the above context, answer the following:
1) What kind of combination exists between M and O?
2)How many electrons are there in the outermost shell of M?
3) Name the group to which M belongs.
4) State the reaction taking place at the cathode.
5) Name the product at the anode.
Which one of the methods given in column I is applied for the extraction of each of the metals given in column II:
| Column I | Column II |
| Electrolytic reduction | Aluminium |
| Reduction with Carbon | Zinc |
| Reduction with Aluminium | Sodium |
| Iron | |
| Manganese | |
| Tin |
Name two metals which occur in nature in free state as well as in combined state.
How is aluminium metal extracted? Explain with the help of an equation.
Which metal is extracted from bauxite ore?
Which of the following reactants are used to carry out the thermite reaction required for welding the broken railway tracks?
(a) Al2O3 + Fe
(b) MnO2 + Al
(c) Fe2O3 + Al
(d) Cu2O + Fe
Distinguish between ‘roasting’ and ‘calcination’. Which of these two is used for sulphide ores and why?
Define the term:
Flux
Complete the incomplete statement with missing word:
Metals form positive ions while non- metals ______.
X is an element in the form of a powder. X burns in oxygen and the product is soluble in water. The solution is tested with litmus. Write down only the word which will correctly complete each of the following sentences:
(a) If X is a metal, then the litmus will turn ______.
(b) If X is a non-metal, then the litmus will turn ______.
(c) If X is a reactive metal, then ______ will be evolved, when X reacts with dilute sulphuric acid.
(d) If X is a non -metal, it will form _ oxide, which will form ______ solution with water.
(e) If X is a non -metal, it will not conduct electricity unless it is carbon in the form of ______.
State the property of the metal being utilized in the following :
| Use of metal | Property |
| Zinc in Galvanization | |
| Aluminium in Thermite welding |
Which one of the following is not true of metal :
Metal will have 1 or 2 or 3 electrons in their valence shell.
Heating an ore in a limited supply of air or in the absence of air at a temperature just below its melting point is known as ______.
Name the following:
A steel grey coloured solid non-metal.
In electrolytic reduction of alumina _______ is used as a cathode.
Write the name.
Metals which are amphoteric in nature.
Explain concept with example/explain with the help of a balanced equation.
Calcination
Which of the following metals exist in their native state in nature?
- Cu
- Au
- Zn
- Ag
An alloy is
