Advertisements
Advertisements
प्रश्न
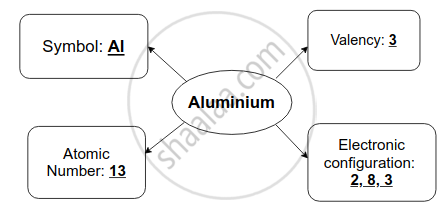
Complete flow chart given below.

Advertisements
उत्तर
संबंधित प्रश्न
What chemical process is used for obtaining a metal from its oxide?
How is manganese extracted from manganese dioxide, MnO2? Explain with the help of an equation.
Explain giving one example, how highly reactive metals (which are high up in the reactivity series) are extracted.
Define the term mineral.
What is the difference between a mineral and an ore?
Name one ore of sodium. Name the sodium compound present in this ore and write its chemical formula.
Which of the following is an iron ore?
(a) cinnabar
(b) calamine
(c) haematite
(d) rock salt
The metal which can be extracted from the bauxite ore is:
(a) Na
(b) Mn
(c) Al
(d) Hg
The metal which can be extracted simply by heating the cinnabar ore in air is:
(a) Zn
(b) Cu
(c) Al
(d) Hg
Which metal can be extracted from the following ore?
bauxite
Give the principles of the hydrolytic method.
Some metallic oxides can be reduced by hydrogen, carbon and carbon monoxide and some cannot. Explain.
Give reasons, why aluminum is used in:
making alloys
Give reasons, why aluminum is used in:
In making ships
Name the following:
The process of heating a substance very strongly in such a way that it does not combine with oxygen.
How are metals refined by the electrolytic methods?
Explain the following reaction with the balanced equation.
Chlorine dissolved in water
Explain in brief types of extraction of highly reactive metals according to their reactivity.
Explain in brief types of extraction of less reactive metals according to their reactivity.
