Advertisements
Advertisements
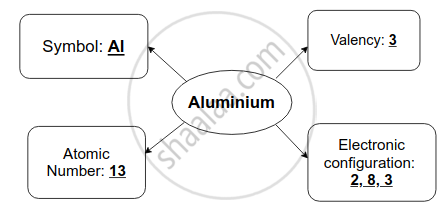
प्रश्न
Complete flow chart given below.

Advertisements
उत्तर
संबंधित प्रश्न
A man went door to door posing as a goldsmith. He promised to bring back the glitter of old and dull gold ornaments. An unsuspecting lady gave a set of gold bangles to him which he dipped in a particular solution. The bangles sparkled like new but their weight was reduced drastically. The lady was upset, but after a futile argument, the man beat a hasty retreat. Can you play the detective to find out the nature of the solution he had used?
In the extraction of aluminium: Draw the diagram for the extraction of aluminium.
Name one metal which is extracted by electrolytic reduction.
A zinc ore on heating in air forms sulphur dioxide. Describe briefly any two stages involved in the conversion of this concentrated ore into zinc metal.
Define the term ore.
The metal which is always present in an amalgam is:
(a) iron
(b) aluminium
(c) mercury
(d) magnesium
Name the following:
Name two metals always find in combined state.
Explain the following:
Galvanization protects iron from rusting.
How many valence electrons are present in non- metals?
How is the method of extraction of metals high up in the reactivity series different from that for metals in the middle? Why can the same process not be applied for them? Name the process used for the extraction of these metals.
Atomic number of aluminium is _______ and its electronic configuration is _______.
Sulphide ores : Roasting : : Carbonate ores : _______
In the electrolytic method, a layer of highly active metal is applied to a less active metal.
Explain in brief types of extraction of highly reactive metals according to their reactivity.
Explain in brief types of extraction of moderately reactive metals according to their reactivity.
Explain Bayer’s process of concentration of bauxite with a chemical equation.
Answer the questions on the following passage.
The minerals from which the metal can be separated economically are called ores. Ores contain many types of impurities such as soil, sand and rocky substances along with metal compounds. These impurities are called gangue.
Metals can be extracted from their ores by means of various methods of separation. The process of extraction of metal in a pure state from the ores is also a part of metallurgy.
Ores are taken out from the mines and the gangue is usually separated from the ore at the site itself by various methods. Then the ores are carried out to the place where metals are produced. Here metals are extracted in pure form. Then metals are further purified by different methods of purification. This entire process is called metallurgy. Most metals being reactive do not occur in nature in the free state but are found in combined state as their salts such as oxides, carbonates, sulphides, and nitrates. however, the most unreactive metals that are not affected by air, water and other natural factors like silver, gold, platinum, generally occur in a free state. The compounds of metals that occur in nature along with the impurities are called minerals.
- What are ores?
- Which processes are involved in the branch of metallurgy? What is metallurgy?
- Which metals are found in a free state?
- In what forms are metals found in combined state?
- What is gangue?
Which of the following ore is concentrated by the Froth floatation process?
2 mL each of concentrated HCl, HNO3 and a mixture of concentrated HCl and concentrated HNO3 in the ratio of 3 : 1 were taken in test tubes labelled as A, B and C. A small piece of metal was put in each test tube. No change occurred in test tubes A and B but the metal got dissolved in test tube C respectively. The metal could be
Give the reaction involved during extraction of zinc from its ore by
- roasting of zinc ore
- calcination of zinc ore
