Advertisements
Advertisements
Question
Answer the following question:
State the four blocks of the modern periodic table based on the electronic configuration of elements.
Advertisements
Solution
Elements in the modern periodic table are classified on the basis of their electronic configuration. They are divided into four blocks: s – block, p – block, d – block, and f – block.
APPEARS IN
RELATED QUESTIONS
Elements in the same group show the same valency. Give scientific reason.
What is meant by 'group' in the modern periodic table? How do the following change on moving from top to bottom in a group?
(i) Number of valence electrons
(ii) Number of occupied shells
(iii) Size of atoms
(iv) Metallic character of element
(v) Effective nuclear charge experienced by valence electrons
Write the number of horizontal rows in the modern periodic table. What are these rows called?
Consider two elements 'A' (Atomic number 17) and 'B' (Atomic number 19) :
(i) Write the positions of these elements in the modern periodic table giving justification.
(ii) Write the formula of the compound formed when 'A' combines with 'B.'
(iii) Draw the electron dot structure of the compound and state the nature of the bond formed between the two elements.
Write any one difference in the electronic configurations of group-1 and group-2 elements ?
Consider two elements 'X' (Atomic number 17) and 'Y' (Atomic number 20)
(i) Write the positions of these elements in the modern periodic table giving justification.
(ii) Write the formula of the compound formed by the combination of 'X' and 'Y'.
(iii) Draw the electron-dot structure of the compound formed and state the nature of the bond formed between the two elements ?
An element with the atomic number 19 will most likely combine chemically with the element whose atomic number is ______.
An element Z has atomic number 16. Answer the following questions on Z:
1) State the period and group to which Z belongs
2) Is Z a metal or a non-metal?
3) State the formula between Z and hydrogen
4) What kind of a compound is this?
What happens to the metallic character of the elements as we go down in a group of the periodic table?
Name the element which is in first group and third period.
How does the electropositive character of elements change on going down in a group of the periodic table?
How does the atomic size vary on going down from top to bottom in a group of the periodic table? Why does it vary this way?
In which part of a group would you separately expect the elements to have the greatest metallic character
In each of the following pair, choose the atom having the bigger size:
Na (At. No. 11) or K (At. No. 19)
How does the number of:
valence electrons and
An element barium has atomic number 56. Look up its position in the periodic table and answer the following question.
Is it more or less reactive than calcium?
An element X belong to 3rd periods and group II of the periodic table state:
the number of valence electrons,
Four elements P, Q, R and S belong to the third period of the Modern Periodic Table and have respectively 1, 3, 5 and 7 electrons in their outermost shells. Write the electronic configurations of Q and R and determine their valences. Write the molecular formula of the compound formed when P and S combine.
Study the extract of the Periodic Table given below and answer the questions that follow. Give the alphabet corresponding to the element in question. DO NOT repeat an element. 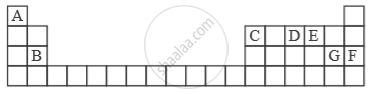
The ion of which element will migrate towards the cathode during electrolysis?
What is meant by periodicity of elements?
Select the correct answer
Most reactive character among the elements given below is found in
Select the correct answer
Identify the metalloid
What is meant by a group in the periodic table?
The following table represents the first period of the modern periodic table. Study the table and answer the questions that follow:
- Write the formula of the sulphate of the element with atomic number 13.
- What type of bonding will be present in the oxide of the element with atomic number 1?
- Which feature of the atomic structure accounts for the similarities in the chemical properties of the elements in group VIIA of the periodic table?
- Name the element which has the highest ionization potential.
- How many electrons are present in the valence shell of the element with atomic number 18?
- What is the name given to the energy released, when an atom in its isolated gaseous state accepts an electron to form an anion?
- What is the electronic configuration of the element in the third period which gains one electron to become an anion?
- Fill in the blanks:
The atomic size ______ as we move from left to right across the period, because the ______ increases, but the ______ remains the same.
The elements of one short period of the periodic table are given below in order from left to right:
Li Be B C O F Ne
(a) To which period do these elements belong?
(b) One element of this period is missing. Which is the missing element and where should it be placed?
(c) Place the three elements, fluorine, beryllium and nitrogen, in the order of increasing electro negativity.
(d) Which one of the above elements belongs to the halogen series?

In the above table, H does not represent hydrogen.Some elements are given in their own symbol and position in the periodic table while others are shown with a letter. With refrence to the table answer the following questions.
1. Identify the most electronegative element.
2. Identify the most reactive element of Group I.
3. Identify the element from Period 3 with least atomic size.
4. How many valence electrons are present in Q?
5. Which element from group 2 would have the least ionisation energy?
6. Identify the noble gas of the fourth period.
7. In the compound between A and H, what type of bond would be formed and give its molecular formula.
Arrange the following as per instruction given in the bracket.
Cl, F, Br, I (increasing electron affinity)
Arrange the following as per instruction given in the bracket.
K, Pb, Ca, Zn (increasing reactivity)
Arrange the following in order of increasing radii:
N, O, P
An element belongs to the third period and Group IIIA (13) of the periodic table. State: the name of the element.
(a) Fill in the blanks and rewrite the completed statements:
(i) ________ group in the periodic table contains elements that are all gases at room temperature.
(ii) Very fine particles mainly scatter ______ light.
Based on the group valency of element write the molecular formula of the following compound giving justification:
Compound formed when an element A of group 2 combines with an element B of group seventeen.
Name the elements in Period 1.
Name or state following with reference to the element of the first three periods of the periodic table.
A metal in period 3 having valency 3.
Fill in the blanks from the words A to F given below.
A: Decreases
B: Increases
C: Remains same
D: Increases by one
E: Electropositive
F: Electronegative
Down a group in the Modern Periodic Table.
No. of electron shells __________; No. of valence electrons ________; Electronegativity ________ Character of elements changes from _________ to ____________.
There are three elements E, F, G with atomic numbers 19, 8 and 17 respectively.
Classify the above elements as metals and non-metals.
