Advertisements
Advertisements
Question
Answer the following in brief.
How will you represent the zeroth-order reaction graphically?
Advertisements
Solution
Graphical representation of zero-order reactions: The rate law is given as
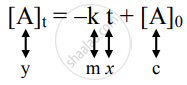
which is a straight line given by y = mx + c.
A plot of `["A"]_"t"` versus t is a straight line.

[A]t vs t for zero-order reaction
The slope of the straight line is -k and its intercept on the y-axis is `["A"]_0`.
APPEARS IN
RELATED QUESTIONS
Answer the following in one or two sentences.
Write the relationships between rate constant and half-life of the first order and zeroth-order reactions.
Answer the following in brief.
Derive the integrated rate law for the first-order reaction.
Time required for 100% completion of a zero order reaction is _______.
Rate constant of a reaction is 3.6 × 10–3 s–1. The order of reaction is ______.
For first order reaction, the rate constant for the decomposition of N2O5 is 6 × 10–4 s –1. The half-life period for decomposition in seconds is ______.
For the reaction 2NOBr → 2NO2 + Br2, the rate law is rate = k[NOBr]2. If the rate of a reaction is 6.5 × 10–6 mol L–1 s–1, when the concentration of NOBr is 2 × 10–3 mol L–1. What would be the rate constant of the reaction?
For a first-order reaction \[\ce{A -> B}\] the rate constant is x min−1. If the initial concentration of A is 0.01 M, the concentration of A after one hour is given by the expression.
For a first order reaction \[\ce{A ->Product}\] with initial concentration x mol L−1, has a half life period of 2.5 hours. For the same reaction with initial concentration `("x"/2)` mol L−1 the half life is

This reaction follows first-order kinetics. The rate constant at particular temperature is 2.303 × 10−2 hour−1. The initial concentration of cyclopropane is 0.25 M. What will be the concentration of cyclopropane after 1806 minutes? (log 2 = 0.3010)
Write the rate law for the following reaction.
A reaction that is `3/2` order in x and zero order in y.
A zero order reaction is 20% complete in 20 minutes. Calculate the value of the rate constant. In what time will the reaction be 80% complete?
A first order reaction is 40% complete in 50 minutes. Calculate the value of the rate constant. In what time will the reaction be 80% complete?
What is the value of rate constant of first order reaction, if it takes 15 minutes for consumption of 20% of reactants?
How long would it take to electroplate a spoon with 0.1 mol of silver (108 g/mol) at a constant current of 2.0 A using AgNO3?
The time of completion of 90% of a first order reaction is approximately ____________.
If [A] is the concentration of A at any time t and [A]0 is the concentration at t = 0, then for the 1st order reaction, the rate equation can be written as ____________.
A first order reaction completes its 10% in 20 minutes, then the time required to complete its 19% is ____________.
In a first order reaction, the concentration of the reactant is reduced to 25% in one hour. The half-life period of the reaction is ____________.
What is the order of reaction if the unit of rate constant (k) is mol dm−3 s−1?
The rate and the rate constants of the reaction, \[\ce{A + 2B -> C + D}\] are 8 × 10−3 mol dm−3 s−1 and 2 × 10−3 s−1 respectively. The concentrations of A and B are 4 mol dm−3 each. The overall order of the reaction is ____________.
For a zero order reaction, the plot of [A]t vs t is linear. The slope of the line is equal to ____________.
The initial concentration of reactant (A) is 2 mol dm−3 for a zero order reaction \[\ce{A -> B}\]. The rate constant (k) is related to its half-life `("t"_(1//2))` by the equation:
Which among the following is an example of pseudo first order reaction?
The rate constant for a first order reaction is 7.0 × 10-4 s-1. If initial concentration of reactant is 0.080 M, what is the half-life of reaction?
Calculate half-life of a first order reaction in minute if the rate constant is 1 × 10-3 s-1.
Which is the relation between half life and rate constant for a zero order?
The half-life of first order reaction is 1.7 hour. What is its rate constant?
The half-life period for the first order reaction is 1.7 hrs. How long will it take for 20% of the reactant to disappear?
