Advertisements
Advertisements
प्रश्न
Answer the following in brief.
How will you represent the zeroth-order reaction graphically?
Advertisements
उत्तर
Graphical representation of zero-order reactions: The rate law is given as
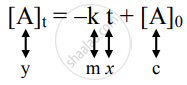
which is a straight line given by y = mx + c.
A plot of `["A"]_"t"` versus t is a straight line.

[A]t vs t for zero-order reaction
The slope of the straight line is -k and its intercept on the y-axis is `["A"]_0`.
APPEARS IN
संबंधित प्रश्न
Answer the following in one or two sentences.
How do the half-lives of the first order and zero-order reactions change with the initial concentration of reactants?
How will you represent first order reactions graphically.
Answer the following in brief.
Derive the integrated rate law for the first-order reaction,
\[\ce{A_{(g)} -> B_{(g)} + C_{(g)}}\] in terms of pressure.
Solve
A first-order reaction takes 40 minutes for 30% decomposition. Calculate its half-life.
Answer the following in brief.
What are the units of the rate constant?
Answer the following in brief.
Give one example and explain why it is pseudo-first-order.
Write order of the following reaction:
\[\ce{2NH_{3(g)} -> N_{2(g)} + 3H_{2(g)}}\]
What is half life of first order reaction if time required to decrease concentration of reactants from 0.8 M to 0.2 M is 12 hours?
Explain with the help of a potential energy diagram that the catalyst increases the rate of the reaction.
Derive an expression for the relation between half-life and rate constant for first-order reaction.
For a first-order reaction \[\ce{A -> B}\] the rate constant is x min−1. If the initial concentration of A is 0.01 M, the concentration of A after one hour is given by the expression.
Give two examples for zero order reaction.
A first order reaction has a rate constant 0.00813 min-1. How long will it take for 60% completion?
A reaction that is of the first order with respect to reactant A has a rate constant 6 min−1. If we start with [A]0 = 0.5 mol dm−3, when would [A] reach the value 0.05 mol dm−3?
A first order reaction completes its 10% in 20 minutes, then the time required to complete its 19% is ____________.
In a first order reaction, the concentration of the reactant, decreases from 0.8 mol dm−3 to 0.4 mol dm−3 in 15 minutes. The time taken for the concentration to change from 0.1 mol dm−3 to 0.025 mol dm−3 is ____________.
The order of the reaction for which the unit of rate constant is s−1 is ____________.
The rate of formation of B at time t for reaction \[\ce{2A -> 3B}\] is equal to ____________.
For a first order reaction, \[\ce{A -> B}\], if [A] = 1 M and rate is 4 × 10−2 M s−1. What is the rate constant of the reaction?
Which among the following is an example of pseudo first order reaction?
A first order reaction takes 40 minutes for 30% completion. Calculate the half-life of reaction.
Half-life of first-order reaction \[\ce{X -> Y + Z}\] is 3 minutes. What is the time required to reduce the concentration of 'X' by 90 % of it's initial concentration?
If the rate constant for a first-order reaction is k, the time (t) required for the completion of 99% of the reaction is given by:
Consider the following reaction.
\[\ce{SO2(g) + 1/2 O2(g) <=>[K1] SO3(g)}\]
\[\ce{2SO3(g)<=>[K2] 2SO2(g) + O2(g)}\]
What is the relation between K1 and K2?
What is the rate constant of a first order reaction if 0.08 mole of reactant reduces to 0.02 mole in 23.03 minutes?
What are integrated rate laws?
The half-life period for the first order reaction is 1.7 hrs. How long will it take for 20% of the reactant to disappear?
