Advertisements
Advertisements
प्रश्न
How will you represent first order reactions graphically.
Advertisements
उत्तर
i. The differential rate law for the first-order reaction A → P is 
The equation is of the form y = mx + c. A plot of rate versus [A]t is a straight line passing through the origin. The slope of straight line = k.
 |
| Variation of rate with [A] |
ii. The integrated rate law is
k = `2.303/t log_10 ["A"]_0/["A"]_"t"`
On rearrangement, the equation becomes
`(kt)/2.303 = log_10 ["A"]_0 - log_10 ["A"]_"t"`
Hence,
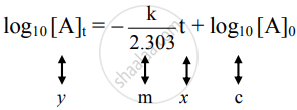
The equation is of the straight line. A graph of `log_10[A]_t` versus t yields a straight line with slope `-"k"/2.303` and y-axis intercepts as log10[A]0.
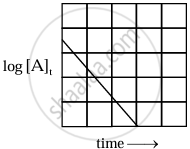 |
| Variation of `log_10 [A]_t` with time |
iii. Rearranging the integrated rate law equation, we get
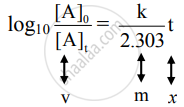
The equation has a straight-line form y = mx. Hence, the graph of `log_10 ([A]_0)/([A]_t)` versus t is a straight line passing through the origin.
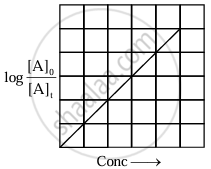 |
| Variation of `log_10 ([A]_0)/([A]_t)` with time |
APPEARS IN
संबंधित प्रश्न
Answer the following in one or two sentences.
How do the half-lives of the first order and zero-order reactions change with the initial concentration of reactants?
In a first-order reaction, the concentration of the reactant decreases from 20 mmol dm−3 to 8 mmol dm−3 in 38 minutes. What is the half-life of reaction?
Time required for 100% completion of a zero order reaction is _______.
Write order of the following reaction:
\[\ce{2NH_{3(g)} -> N_{2(g)} + 3H_{2(g)}}\]
Explain pseudo first order reaction with a suitable example.
The decomposition of phosphine (PH3) on tungsten at low pressure is a first-order reaction. It is because the
The rate constant of a reaction is 5.8 × 10−2 s−1. The order of the reaction is ____________.
Give two examples for zero order reaction.
The time for half change in a first order decomposition of a substance A is 60 seconds. Calculate the rate constant. How much of A will be left after 180 seconds?
From the following data, show that the decomposition of hydrogen peroxide is a reaction of the first order:
| t (min) | 0 | 10 | 20 |
| V (ml) | 46.1 | 29.8 | 19.3 |
Where t is the time in minutes and V is the volume of standard KMnO4 solution required for titrating the same volume of the reaction mixture.
A first order reaction is 40% complete in 50 minutes. Calculate the value of the rate constant. In what time will the reaction be 80% complete?
How long would it take to electroplate a spoon with 0.1 mol of silver (108 g/mol) at a constant current of 2.0 A using AgNO3?
For first order reaction the concentration of reactant decreases from 0.2 to 0.1 M in 100 minutes. What is the rate constant of the reaction?
A first order reaction completes its 10% in 20 minutes, then the time required to complete its 19% is ____________.
The rate and the rate constants of the reaction, \[\ce{A + 2B -> C + D}\] are 8 × 10−3 mol dm−3 s−1 and 2 × 10−3 s−1 respectively. The concentrations of A and B are 4 mol dm−3 each. The overall order of the reaction is ____________.
Half-life period of a first order reaction, \[\ce{A -> product}\] is 3.0 hours. What is the value of rate constant?
A first order reaction is 75% completed in 60 minutes, the time required for it's 50% completion is ____________.
For a zero order reaction, the plot of [A]t vs t is linear. The slope of the line is equal to ____________.
The half-life of a first order reaction is 6.0 hour. How long will it take for the concentration of reactant to decrease from 0.4 M to 0.12 M?
Which among the following is an example of zero order reaction?
For a first order reaction, \[\ce{A -> B}\], if [A] = 1 M and rate is 4 × 10−2 M s−1. What is the rate constant of the reaction?
A first order reaction takes 40 minutes for 30% completion. Calculate the half-life of reaction.
Half-life of first order reaction is 20 minutes. What is the time taken to reduce the initial concentration of the reactant to `1/10`th?
Half-life of first-order reaction \[\ce{X -> Y + Z}\] is 3 minutes. What is the time required to reduce the concentration of 'X' by 90 % of it's initial concentration?
The integrated rate law is a direct relationship between time and ______
If the rate constant for a first-order reaction is k, the time (t) required for the completion of 99% of the reaction is given by:
Which of the following is correct for a first-order reaction?
A radioactive isotope decayed to 17/32 of its original mass after 60 minutes. Find the half-life of this radioisotope.
