Advertisements
Advertisements
Question
Answer in brief.
Write reaction showing conversion of Acetaldehyde into acetaldehyde dimethyl acetal.
Advertisements
Solution
Step 1:
\[\begin{array}{cc}
\ce{H3C\phantom{..............................}}\ce{H\phantom{.........}}\\
|\phantom{..................................}|\phantom{.........}\\
\phantom{.}\ce{C = O + CH3 - OH ⇌[dry HCl][dil.HCl] H3C - C - OCH3}\\
\phantom{.}|\phantom{..................................}|\phantom{.........}\\
\phantom{..}\ce{\underset{Acetaldehyde}{H}}\phantom{......................}\ce{\underset{Hemiacetal (unstable)}{OH}}\phantom{........}
\end{array}\]
Step 2:
\[\begin{array}{cc}
\ce{H\phantom{.....................................}}\ce{H\phantom{.........}}\\
|\phantom{......................................}|\phantom{.........}\\
\ce{H3C - C - OCH3 + CH3 - OH ⇌[dry HCl][dil.HCl] H3C - C - OCH3 + H2O}\\
|\phantom{......................................}|\phantom{.........}\\
\ce{\underset{Hemiacetal (unstable)}{H}\phantom{....................}}
\ce{\underset{Acetaldehyde dimethyl acetal (stable)}{OCH3}\phantom{.}}
\end{array}\]
RELATED QUESTIONS
Write reactions for the action of the following reagents on p-chlorobenzaldehyde.
Phenyl hydrazine
If acetaldehyde is treated with Fehling's solution, the change that occurs in the system is ____________.
Compounds of general formula, are called ____________.
\[\begin{array}{cc} \phantom{...}\ce{R}\phantom{....}\ce{OR''}\phantom{}\\ \phantom{}\backslash\phantom{..}/\\ \ce{C}\\ \phantom{}/\phantom{..}\backslash\\ \phantom{...}\ce{R'}\phantom{...}\ce{OR''}\phantom{} \end{array}\]
Acetone on Clemmensen reduction gives ____________.
Which of the following will give positive iodoform test?
Which of the following is Clemmensen reduction?
Which of the following reagents is used for the following conversion?
\[\ce{CH3 - CH = CH - CHO -> CH3 - CH = CH - CH2OH}\]
Which one of the following reaction is an example of disproportionation reaction.
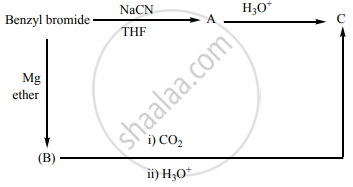
Identify X and Y.
\[\ce{CH3COCH2CH2COOC2H5 ->[CH3MgBr] X ->[H3O^+] Y}\]
Identify A, B, C and D.
\[\ce{ethanoic acid ->[SOCl2] A ->[Pd/BaSO4] B ->[NaOH] C ->[][\Delta] D}\]
What is the action of HCN on ethanal?
Identify A, B and C.
How will you prepare acetic anhydride from acetic acid?
How will you prepare acetamide from methyl cyanide?
How will you prepare lactic acid from ethanol?
How will you prepare cinnamic acid from benzaldehyde?
Which of the following compound will show a positive silver mirror test?
Which of the following compounds will undergo self-aldol condensation in the presence of cold dilute alkali?
Which of the following reaction does not involve either oxidation or reduction?
Aldehydes are readily oxidised to yield carboxylic acids but ketones are inert to oxidation. Which is the most likely explanation regarding this difference in reactivity?
Write the structure of the major product of the aldol condensation of benzaldehyde with acetone.
Write the structure of the major product of the aldol condensation of benzaldehyde with acetone.
Write a note on Clemmensen reduction.
Identify B in the following reaction:
\[\begin{array}{cc}
\ce{O}\phantom{........}\\
||\phantom{........}\\
\ce{\underset{(Acetone)}{CH3 - C - CH3}->[CrO3] B}
\end{array}\]
Write reactions when phenol reacts with Dilute HNO3
Write the structure of the major product of the aldol condensation of benzaldehyde with acetone.
How will you prepare Acetic anhydride from acetic acid ?
