Advertisements
Advertisements
Question
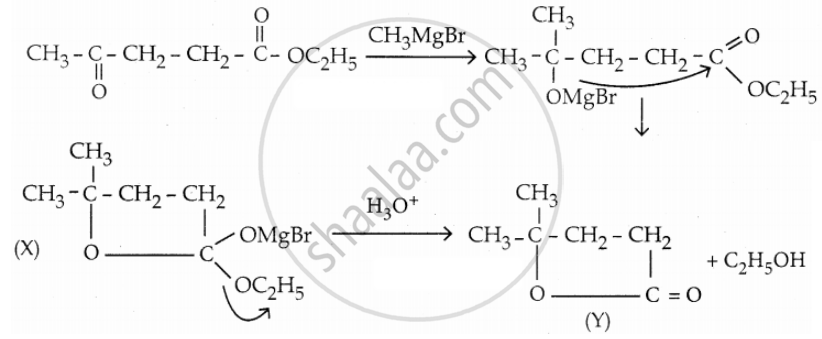
Identify X and Y.
\[\ce{CH3COCH2CH2COOC2H5 ->[CH3MgBr] X ->[H3O^+] Y}\]
Advertisements
Solution
APPEARS IN
RELATED QUESTIONS
Write reactions for the action of the following reagents on p-chlorobenzaldehyde.
Phenyl hydrazine
Aldol condensation between which of the following compounds followed by dehydration gives 4-Methylpent-3-en-2-one?
How is the following conversion effected propanal into butanone?
How is the following conversion effected Hex-3-yne into hexan-3-one?
How is the following conversion effected phenyl methanal into benzoic acid?
Which one of the following substituents at para-position is most effective in stabilizing the phenoxide 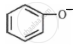 ion?
ion?
Write a note on Clemmensen reduction.
Write reactions when phenol reacts with Dilute HNO3
How will you prepare Acetic anhydride from acetic acid ?
Write the structure of the major product of the aldol condensation of benzaldehyde with acetone.
