Advertisements
Advertisements
Question
Answer in brief.
Observe the following equation of reaction of Tollens' reagent with aldehyde. How do we know that a redox reaction has taken place? Explain.
\[\ce{R-CHO + 2Ag(NH_{3})^{+}_{2} + OH^{-}->[\triangle] R-CO{O}^{-} + 2Ag↓ + 4NH3 + 2H2O}\]
Advertisements
Solution
When an aldehyde is boiled with Tollens' reagent, it gets oxidized to carboxylate ion and Ag+ ion is reduced to silver (Ag). The oxidation number of carbon in aldehyde increases while the oxidation number of Ag decreases. Hence, this is a redox reaction.
APPEARS IN
RELATED QUESTIONS
Answer in brief.
What is the action of hydrazine on cyclopentanone in presence of KOH in ethylene glycol?
Write a note on Stephen reaction.
Write reaction showing aldol condensation of cyclohexanone.
_____________ reaction is used to synthesize straight alkyl substituted benzenes.
The following compounds will give positive Fehling’s test.
Write another name of disproportionation reaction?
Explain haloform reaction with suitable example.
Aldol condensation between which of the following compounds followed by dehydration gives 4-Methylpent-3-en-2-one?
Benzaldehyde gives all tests positive EXCEPT ____________.
Acetone reacts with iodine (I2) to form iodoform, in the presence of ____________.
Which of following is not a property of red phosphorus?
Which of the following does not give yellow solid on treatment with sodium hypoiodlte?
Acetone on Clemmensen reduction gives ____________.
Predict the product Z in the following series of reactions.
\[\ce{Ethanoic acid ->[PCl5] X ->[C6H6][Anhydrous AlCl3] Y ->[i) CH3MgBr][H3O^+] Z}\]
\[\ce{Benzoic acid ->[i) NH3][ii) \Delta] A ->[NaOBr] B ->[NaNO2/HCl] C}\] ‘C’ is:
Which one of the following reaction is an example of disproportionation reaction.
Identify X and Y.
\[\ce{CH3COCH2CH2COOC2H5 ->[CH3MgBr] X ->[H3O^+] Y}\]
How will you convert benzaldehyde into the following compound?
Benzoic acid
How is the following conversion effected propanal into butanone?
Complete the following reaction.
\[\begin{array}{cc}
\ce{CH3 - CH2 - CH2 - C - CH3 ->[HO - CH2 - CH2 - CH2 - OH][dry HCl] ?}\\
||\phantom{.........}\\
\ce{O}\phantom{.........}
\end{array}\]
How will you prepare acetophenone from acetyl chloride?
The reagent used in Wolf - Kishner reduction is ______.
Which one of the following substituents at para-position is most effective in stabilizing the phenoxide 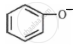 ion?
ion?
Which of the following compounds does not undergo a haloform reaction?
Aldehydes are readily oxidised to yield carboxylic acids but ketones are inert to oxidation. Which is the most likely explanation regarding this difference in reactivity?
Write the structure of the major product of the aldol condensation of benzaldehyde with acetone.
What is the action of the following regent on propanal?
Sodium Bisulphite
Write the structure of the major product of the aldol condensation of benzaldehyde with acetone.
Write the structure of the major product of the aldol condensation of benzaldehyde with acetone.
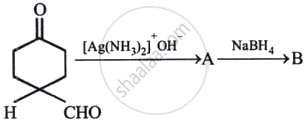
The products formed in the following reaction, A and B are: