Advertisements
Advertisements
प्रश्न
Answer in brief.
Observe the following equation of reaction of Tollens' reagent with aldehyde. How do we know that a redox reaction has taken place? Explain.
\[\ce{R-CHO + 2Ag(NH_{3})^{+}_{2} + OH^{-}->[\triangle] R-CO{O}^{-} + 2Ag↓ + 4NH3 + 2H2O}\]
Advertisements
उत्तर
When an aldehyde is boiled with Tollens' reagent, it gets oxidized to carboxylate ion and Ag+ ion is reduced to silver (Ag). The oxidation number of carbon in aldehyde increases while the oxidation number of Ag decreases. Hence, this is a redox reaction.
APPEARS IN
संबंधित प्रश्न
Answer in brief.
Write reaction showing conversion of Acetaldehyde into acetaldehyde dimethyl acetal.
Write reactions for the action of the following reagents on p-chlorobenzaldehyde.
Phenyl hydrazine
Which of the following is Schiff's reagent?
Ethyl methyl ketone can be reduced to n-butane by which of the following reactions?
Identify the INCORRECT reaction.
If acetaldehyde is treated with Fehling's solution, the change that occurs in the system is ____________.
Acetone reacts with iodine (I2) to form iodoform, in the presence of ____________.
Which of the following represents the structure of isopropyl methyl ketone?
Which of the following does not give yellow solid on treatment with sodium hypoiodlte?
Fehling solution is ____________.
In Clemmensen's reaction, ketone gives ____________.
Which of the following will give positive iodoform test?
Which of the following reagents is used to avoid further oxidation of aldehydes?
Which of the following reagents is used for the following conversion?
\[\ce{CH3 - CH = CH - CHO -> CH3 - CH = CH - CH2OH}\]
The formation of cyanohydrin from acetone is an example of ____________.
Reaction of acetone with one of the following reagents involves nucleophilic addition followed by elimination of water. The reagent is:
\[\ce{Ethanoic acid ->[P/Br2] 2-bromoethanoic acid}\]. This reaction is called ____________.
Identify A, B, C and D.
\[\ce{ethanoic acid ->[SOCl2] A ->[Pd/BaSO4] B ->[NaOH] C ->[][\Delta] D}\]
How will you prepare acetamide from methyl cyanide?
How will you prepare ethane from sodium acetate?
Which one of the following reactions will not result in the formation of a carbon-carbon bond?
Which of the following is most reactive towards the addition reaction of hydrogen cyanide to form the corresponding cyanohydrin?
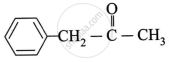
Write the structure of the major product of the aldol condensation of benzaldehyde with acetone.
Write a note on Clemmensen reduction.
Write the structure of the products obtained from the following ketones by action of hydrazine in presence of strong base KOH.
Write the structure of the major product of the aldol condensation of benzaldehyde with acetone.
How will you prepare Acetic anhydride from acetic acid.
How will you prepare acetic anhydride from acetic acid?