Advertisements
Advertisements
Question
How will you prepare ethane from sodium acetate?
Advertisements
Solution
Conversion of sodium acetate into ethane:
\[\ce{\underset{(Sodium acetate)}{2CH3COONa} + ->[Electrolysis] \underset{(Ethane)}{CH3 - CH3} + 2CO2 + 2Na}\]
APPEARS IN
RELATED QUESTIONS
Write the name of the product when ketones react with 1,2-diol in presence of dry HCl.
Write reactions for the action of the following reagents on p-chlorobenzaldehyde.
Tollen’s reagent
Aldol condensation between which of the following compounds followed by dehydration gives 4-Methylpent-3-en-2-one?
Which of the following is Schiff's reagent?
Compounds of general formula, are called ____________.
\[\begin{array}{cc} \phantom{...}\ce{R}\phantom{....}\ce{OR''}\phantom{}\\ \phantom{}\backslash\phantom{..}/\\ \ce{C}\\ \phantom{}/\phantom{..}\backslash\\ \phantom{...}\ce{R'}\phantom{...}\ce{OR''}\phantom{} \end{array}\]
How will you prepare ethyl acetate from methyl acetate?
How will you prepare acetaldehyde from ethyne?
Write reactions when phenol reacts with Dilute HNO3
Write the structure of the major product of the aldol condensation of benzaldehyde with acetone.
What is IUPAC name for
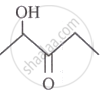 ?
?
