Advertisements
Advertisements
प्रश्न
How will you prepare ethane from sodium acetate?
Advertisements
उत्तर
Conversion of sodium acetate into ethane:
\[\ce{\underset{(Sodium acetate)}{2CH3COONa} + ->[Electrolysis] \underset{(Ethane)}{CH3 - CH3} + 2CO2 + 2Na}\]
APPEARS IN
संबंधित प्रश्न
Identify the product formed in the reaction.

Carboxylic acids have higher boiling points than aldehydes, ketones and even alcohols of comparable molecular mass. It is due to their ____________.
Identify X and Y.
\[\ce{CH3COCH2CH2COOC2H5 ->[CH3MgBr] X ->[H3O^+] Y}\]
Which of the following compound will show a positive silver mirror test?
Which of the following reaction does not involve either oxidation or reduction?
What is the number of different products formed when mixture of ethanal and propanal reacts with aq. NaOH after warming?
Which of the following compounds does not undergo a haloform reaction?
Write the structure of the products obtained from the following ketones by action of hydrazine in presence of strong base KOH.
Write reactions when phenol reacts with Dilute HNO3
What is IUPAC name for
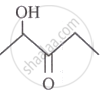 ?
?