Advertisements
Advertisements
Question
Write reactions for the action of the following reagents on p-chlorobenzaldehyde.
Phenyl hydrazine
Advertisements
Solution
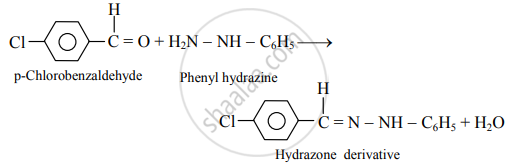
APPEARS IN
RELATED QUESTIONS
Answer in brief.
Observe the following equation of reaction of Tollens' reagent with aldehyde. How do we know that a redox reaction has taken place? Explain.
\[\ce{R-CHO + 2Ag(NH_{3})^{+}_{2} + OH^{-}->[\triangle] R-CO{O}^{-} + 2Ag↓ + 4NH3 + 2H2O}\]
Answer in brief.
Write reaction showing conversion of Acetaldehyde into acetaldehyde dimethyl acetal.
_____________ reaction is used to synthesize straight alkyl substituted benzenes.
Write the name of the product when ketones react with 1,2-diol in presence of dry HCl.
Identify the INCORRECT reaction.
Which of following is not a property of red phosphorus?
Compounds of general formula, are called ____________.
\[\begin{array}{cc} \phantom{...}\ce{R}\phantom{....}\ce{OR''}\phantom{}\\ \phantom{}\backslash\phantom{..}/\\ \ce{C}\\ \phantom{}/\phantom{..}\backslash\\ \phantom{...}\ce{R'}\phantom{...}\ce{OR''}\phantom{} \end{array}\]
In Clemmensen's reaction, ketone gives ____________.
Following reaction is an example of ____________.
\[\ce{Benzaldehyde ->[Nitration] {m}-Nitrobenzaldehyde}\]
Which of the following reagents is used to avoid further oxidation of aldehydes?
Which of the following reagents is used for the following conversion?
\[\ce{CH3 - CH = CH - CHO -> CH3 - CH = CH - CH2OH}\]
\[\ce{Benzoic acid ->[i) NH3][ii) \Delta] A ->[NaOBr] B ->[NaNO2/HCl] C}\] ‘C’ is:
In which case, chiral carbon is not generated by reaction with HCN.
Carboxylic acids have higher boiling points than aldehydes, ketones and even alcohols of comparable molecular mass. It is due to their ____________.
Identify X and Y.
\[\ce{CH3COCH2CH2COOC2H5 ->[CH3MgBr] X ->[H3O^+] Y}\]
How is the following conversion effected phenyl methanal into benzoic acid?
How will you prepare lactic acid from ethanol?
How will you prepare acetaldehyde from ethyne?
How acetone is converted into propane.
How propanal is converted into propane.
Which one of the following substituents at para-position is most effective in stabilizing the phenoxide 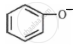 ion?
ion?

In the above reaction, product (B) is:
How are the following compound obtained from alkyne?
Acetaldehyde
Write a note on Clemmensen reduction.
How will you prepare acetic anhydride from acetic acid.
How will you prepare Acetic anhydride from acetic acid.
How will you prepare Acetic anhydride from acetic acid.
How will you prepare acetic anhydride from acetic acid?
