Advertisements
Advertisements
Question
Reaction of acetone with one of the following reagents involves nucleophilic addition followed by elimination of water. The reagent is:
Options
Grignard reagent
Sn/HCl
hydrazine in presence of slightly acidic solution
hydrocyanic acid
Advertisements
Solution
hydrazine in presence of slightly acidic solution
APPEARS IN
RELATED QUESTIONS
Write a note on Stephen reaction.
Write reaction showing aldol condensation of cyclohexanone.
Identify A in the following reaction.
\[\ce{A + CH3MgBr ->[Ether] complex ->[H3O+] (CH3)3C - OH}\]
ln which of the following reactions, both oxidised and reduced forms of the same compound are obtained?
What is the action of HCN on 2, 4-dichlorobenzaldehyde?
How is the following conversion effected Hex-3-yne into hexan-3-one?
How will you prepare malachite green from benzaldehyde?
Which one of the following substituents at para-position is most effective in stabilizing the phenoxide 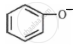 ion?
ion?
Which of the following compound will give positive iodoform test?
What is IUPAC name for
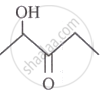 ?
?
