Advertisements
Advertisements
Question
Answer briefly.
What are radio waves?
Advertisements
Solution
- Radio waves are produced by accelerated motion of charges in a conducting wire. The frequency of waves produced by the circuit depends upon the magnitudes of the inductance and the capacitance.
- Thus, by choosing suitable values of the inductance and the capacitance, radio waves of the desired frequency can be produced.
APPEARS IN
RELATED QUESTIONS
What is the range of the wavelength of the following electromagnetic waves?
(a) Micro waves .
Name the region beyond the red end of the spectrum.
What do you understand by the invisible spectrum?
Name two electromagnetic waves of frequency smaller than that of violet light. State one use of each.
Two waves A and B have wavelength 0.01 Å and 9000 Å respectively.
- Name the two waves.
- Compare the speeds of these waves when they travel in vacuum.
What are ultraviolet radiations?
An electromagnetic wave has a frequency of 500 MHz and a wavelength of 60 cm Calculate the velocity of the wave.
In a Coolidge tube, electrons strike the target and stop inside it. Does the target get more and more negatively charged as time passes?
X-ray and visible light travel at the same speed in vacuum. Do they travel at the same speed in glass?
Can a hydrogen atom emit characteristic X-rays?
Why is exposure to X-rays injurious to health but not exposure to visible light, when both are electromagnetic waves?
An X-ray beam can be deflected
Frequencies of Kα X-rays of different materials are measured. Which one of the graphs in the figure may represent the relation between the frequency v and the atomic number Z ?
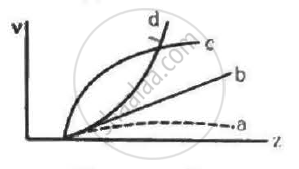
50% of the X-rays coming from a Coolidge tube are able to pass through a 0.1 mm thick aluminium foil. If the potential difference between the target and the filament is increased, the fraction of the X-rays passing through the same foil will be
The Kα X-rays of aluminium (Z = 13) and zinc (Z = 30) have wavelengths 887 pm and 146 pm respectively. Use Moseley's law √v = a(Z − b) to find the wavelengths of the Kα X-ray of iron (Z = 26).
(Use Planck constant h = 6.63 × 10-34 Js= 4.14 × 10-15 eVs, speed of light c = 3 × 108 m/s.)
The energy of a silver atom with a vacancy in K shell is 25.31 keV, in L shell is 3.56 keV and in M shell is 0.530 keV higher than the energy of the atom with no vacancy. Find the frequency of Kα, Kβ and Lα X-rays of silver.
(Use Planck constant h = 6.63 × 10-34 Js= 4.14 × 10-15 eVs, speed of light c = 3 × 108 m/s.)
When 40 kV is applied across an X-ray tube, X-ray is obtained with a maximum frequency of 9.7 × 1018 Hz. Calculate the value of Planck constant from these data.
(Use Planck constant h = 6.63 × 10-34 Js= 4.14 × 10-15 eVs, speed of light c = 3 × 108 m/s.)
Write the range of the wavelength of the following electromagnetic radiations:
(a) Infrared rays
(b) Ultraviolet rays
(c) γ -rays
Write one use of each of the above.
Name the scientist who discovered Visible light
Name the scientist who discovered Ultraviolet rays
Name the part of the electromagnetic spectrum which is:
Suitable for radar systems used in aircraft navigation.
Name the part of the electromagnetic spectrum which is:
Produced by bombarding a metal target with high electrons.
Answer briefly.
Can we produce a pure electric or magnetic wave in space? Why?
Answer briefly.
Does an ordinary electric lamp emit EM waves?
A radio can tune to any station in the 7.5 mHz to 12 MHz band. What is corresponding wave length band.
What is time period of the light for which the eye is most sensitive?
Why does microwave oven heats up a food item containing water molecules most efficiently?
Below is an incomplete table showing the arrangement of electromagnetic spectrum in the increasing order of their wavelength. Complete the table:
| Gamma ray | X - ray | UV rays | Visible rays | Infrared | A | Radio waves |
- Identify the radiation A.
- Name the radiation used to detect fracture in bones.
- Name one property common to both A and Radio waves.
In an atom X, electrons absorb the energy from an external source. This energy “excites” the electrons from a lower-energy level to a higher-energy level around the nucleus of the atom. When electrons return to the ground state, they emit photons.
The figure below is the energy level diagram of atom X with three energy levels, E1 = 0.00eV, E2 = 1.78eV and E3 = 2.95eV. The ground state is considered 0 eV for reference. The transition of electrons takes place between levels E1 and E2.

- What wavelength of radiation is needed to excite the atom to energy level E2 from E1?
- Suppose the external source has a power of 100 W. What would be the rate of photon emission?
