Advertisements
Advertisements
प्रश्न
Write preparation, properties, and uses of Teflon.
Advertisements
उत्तर
Preparation of Teflon:
- The monomer used in the preparation of Teflon is tetrafluoroethylene (CF2 = CF2), which is a gas at room temperature.
- Tetrafluoroethylene is polymerized by using free-radical initiators such as hydrogen peroxide or ammonium persulphate at high pressure to produce polytetrafluoroethylene (Teflon).
\[\ce{\underset{Tetrafluoroethylene}{{n} CF2 = CF2} ->[Polymerization][Peroxide]\underset{Teflon}{[-CF2 - CF2 -]}_{n}}\]
Properties of Teflon:
- Teflon is tough, chemically inert, and resistant to heat and attack by corrosive reagents.
- C – F bond is very difficult to break and remains unaffected by corrosive alkali and organic solvents.
Uses: Teflon is used in making non-stick cookware, oil seals, gaskets, etc.
संबंधित प्रश्न
Write the reactions involved in the preparation of PVC
Explain the following term: Homopolymers
Based on molecular forces, what type of polymer is neoprene?
Draw the structures of veronal and thymine.
Write the monomers of the following polymer :

Answer the following in one sentence.
Define the term: Vulcanization
Answer the following in one sentence.
Identify thermoplastic and thermosetting plastic from the following:
- PET
- Urea formaldehyde resin
- Polythene
- Phenol formaldehyde resin
Name and draw structure of the repeating unit in natural rubber.
Identify condensation polymers and addition polymers from the following.
\[\begin{array}{cc}\ce{-(CH2 - CH -)_{n}}\\
\phantom{....}|\\\ce{\phantom{.......}C6H5}
\end{array}\]
Identify condensation polymers and addition polymers from the following.
-(CH2 - CH = CH - CH2 -)n
Identify condensation polymers and addition polymers from the following.
-(CO(CH2)4 - CONH(CH2)6NH-)n
Identify condensation polymers and addition polymers from the following.
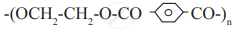
Attempt the following:
Explain the vulcanisation of rubber. Which vulcanizing agents are used for the following synthetic rubber?
a. Neoprene
b. Buna-N
Attempt the following:
What is meant by LDP and HDP? Mention the basic difference between the same with suitable examples.
Answer the following.
Is synthetic rubber better than natural rubber? If so, in what respect?
Nylon 6, 6 is a condensation polymer of hexamethylenediamine and _____________
Write the name of the catalyst used for preparation of high density polythene polymer.
Monomer used for preparation of polyacrylonitrile is _____________
Write a chemical reaction for the preparation of the following polymer.
polyacrylonitrile
Write preparation of low density polythene.
Write the name and formulae of the monomers used for the preparation of dacron.
Which of the following polymer is used in paints?
Identify additional polymers from the following.
I. \[\begin{array}{cc}
\ce{-(CH2 - CH -)_{{n}}}\\
\phantom{....}|\\
\phantom{.......}\ce{C6H5}
\end{array}\]
II. \[\ce{-(CH2 - CH = CH - CH2 -)_{{n}}}\]
III. \[\ce{-(CO(CH2)4 - CONH(CH2)6NH -)_{{n}}}\]
IV.
![]()
Which of the following is the monomer of neoprene?
Which of the following catalysts is used in preparation of terylene?
Which among the following polymers is obtained from CH2 = CH – CN by polymerisation?
Identify the catalyst used in the manufacture of high density polythene.
Which among the following monomers is used to prepare Teflon?
Which of the following polymer is used to make blankets?
Identify addition polymer from the following.
Name the polymers used in laminated sheets and give the name of monomeric units involved in its formation.
Match the polymers given in Column I with their repeating units given in Column II.
| Column I | Column II |
| (i) Acrilan |
(a) \[\begin{array}{cc} |
| (ii) Polystyrene | (b) \[\begin{array}{cc} \ce{Cl}\phantom{.......}\\ |\phantom{........}\\ \phantom{}\ce{-(CH2 - C = CH - CH2)\underset{n}{-}} \end{array}\] |
| (iii) Neoprene | (c) \[\begin{array}{cc} \phantom{................................}\ce{CN}\\ \phantom{..............................}|\\ \ce{-(CH2 - CH = CH - CH2 - CH2 - CH)\underset{n}{-}} \end{array}\] |
| (iv) Novolac | (d) \[\begin{array}{cc} \ce{-(CH2 - CH)\underset{n}{-}}\\ \phantom{.....}|\\ \phantom{.......}\ce{CN} \end{array}\] |
| (v) Buna—N | (e)  |
| (f) \[\begin{array}{cc} \ce{-(CH2 - CH)\underset{n}{-}}\\ \phantom{.....}|\\ \phantom{......}\ce{Cl} \end{array}\] |
Which one of the following polymers are prepared by addition polymerization?
Nylon threads are made of ______.
Which among the following polymers has high tensile strength and is used to obtain tyre cords?
Which of the foolowing polymer is used in the manufacture of insulators.
Answer the following.
Name and draw structure of the repeating unit in natural rubber.
Answer the following.
Write the structure of isoprene and the polymer obtained from it.
Name and draw the structure of the repeating unit in natural rubber.
Another name of terylene is ______.
The monomer used in preparation of teflon is ______.
Name and draw the structure of the repeating unit in natural rubber.
Name and draw structure of the repeating unit in natural rubber.
Write the structure of isoprene and the polymer obtained from it.
Write the structure of isoprene and the polymer obtained from it.
Write the structure of isoprene and the polymer obtained from it.
Identify polyamide polymer from following.
Which among the following is a monomer of natural rubber?
