Advertisements
Advertisements
प्रश्न
Which of the following is most acidic?
पर्याय
Benzyl alcohol
Cyclohexanol
Phenol
m-chlorophenol
Advertisements
उत्तर
m-chlorophenol
Explanation:
m-chlorophenol is most acidic. The alpha carbon of benzyl alcohol and cyclohexanol is sp3 hybridized. In m-chlorophenol, it is sp2 hybridized. In m-chlorophenol, the electron-withdrawing group (−Cl) is present at the meta position.
APPEARS IN
संबंधित प्रश्न
Write IUPAC name of the following compound:
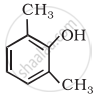
Write IUPAC name of the following compound:
C6H5 – O – C2H5
- Draw the structures of all isomeric alcohols of molecular formula C5H12O and give their IUPAC names.
- Classify the isomers of alcohols in the above question as primary, secondary and tertiary alcohols.
Give IUPAC name of the following ether:
CH3CH2CH2OCH3
Give IUPAC name of the following ether:
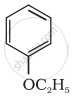
What is the action of hot HI on it?
3-Methylbutane-2-ol on heating with HI gives ______
Write the structures of the products when Butan-2-ol reacts with CrO3
Write the structures of the products when Butan-2-ol reacts with SOCl2
Write the IUPAC name of the following :

Write IUPAC names of the following

Butane-2-ol is ____________.
HBr reacts fastest with ____________.
Which of the following gives a positive iodoform test?
Match the structures of the compounds given in Column I with the name of the compounds given in Column II.
| Column I | Column II | |
| (i) |  |
(a) Hydroquinone |
| (ii) |  |
(b) Phenetole |
| (iii) | 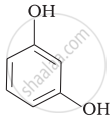 |
(c) Catechol |
| (iv) |  |
(d) o-Cresol |
| (v) |  |
(e) guinone |
| (vi) | 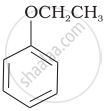 |
(f) Resorcinol |
| (g) Anisole |
Assertion: Like bromination of benzene, bromination of phenol is also carried out in the presence of Lewis acid.
Reason: Lewis acid polarises the bromine molecule.
Convert the following:
Ethyl alcohol into ethyl acetate.
Draw structure of the following compound.
Prop-2-en-1-ol
