Advertisements
Advertisements
Question
Which of the following is most acidic?
Options
Benzyl alcohol
Cyclohexanol
Phenol
m-chlorophenol
Advertisements
Solution
m-chlorophenol
Explanation:
m-chlorophenol is most acidic. The alpha carbon of benzyl alcohol and cyclohexanol is sp3 hybridized. In m-chlorophenol, it is sp2 hybridized. In m-chlorophenol, the electron-withdrawing group (−Cl) is present at the meta position.
APPEARS IN
RELATED QUESTIONS
Write the IUPAC name of the given compound:

Write IUPAC name of the following compound:
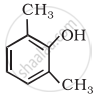
Write structures of the compounds whose IUPAC names are as follows:
3-Chloromethylpentan-1-ol.
- Draw the structures of all isomeric alcohols of molecular formula C5H12O and give their IUPAC names.
- Classify the isomers of alcohols in the above question as primary, secondary and tertiary alcohols.
Give IUPAC name of the following ether:

How is phenol converted into the following?
Benzene
Write the structures of the products when Butan-2-ol reacts with SOCl2
Propanoic acid to ethylamine.
\[\ce{HC ≡ CH ->[HgSO4][H2SO4] ->[CH3MgBr][H2O] ->[PBr3]}\]
The product of acid catalysed hydration of 2-phenylpropene is:
\[\ce{Phenol ->[Zn, dust] 'X' ->[CH3Cl][Anhy. AlCl3] 'Y' ->[Alkaline][KMnO4] 'Z'}\]
The product ‘Z’ is:
Assertion: Like bromination of benzene, bromination of phenol is also carried out in the presence of Lewis acid.
Reason: Lewis acid polarises the bromine molecule.
Write complete reaction for the bromination of phenol in aqueous and non-aqueous medium.
Write the IUPAC name of the following compound.

Convert the following:
Ethyl alcohol into ethyl acetate.
Write IUPAC name of the following compound:
\[\begin{array}{cc}
\phantom{................}\ce{CH3}\\
\phantom{.............}|\\
\ce{CH3 - CH - CH - C - CH3}\\
|\phantom{......}|\phantom{......}|\\
\phantom{...}\ce{CH3}\phantom{..}\ce{OH}\phantom{...}\ce{CH3}
\end{array}\]
Write structural formulae for:
p-Nitrophenol
Write the IUPAC name of the following compound:
\[\begin{array}{cc}
\phantom{..............}\ce{CH3}\\
\phantom{............}|\\
\ce{CH3 - CH - CH - C - CH3}\\
|\phantom{.....}|\phantom{......}|\\
\phantom{...}\ce{CH3}\phantom{.}\ce{OH}\phantom{...}\ce{CH3}
\end{array}\]
