Advertisements
Advertisements
प्रश्न
From the rate expression for the following reaction, determine the order of reaction and the dimension of the rate constant.
\[\ce{C2H5Cl_{(g)} -> C2H4_{(g)} + HCl_{(g)}}\] Rate = k [C2H5Cl]
Advertisements
उत्तर
Given rate = k [C2H5Cl]
Therefore, order of the reaction = 1
Dimension of k = \[\ce{\frac{Rate}{[C2H5Cl]}}\]
= \[\ce{\frac{(mol L^{-1} s^{-1})}{mol L^{-1}}}\]
= s−1
APPEARS IN
संबंधित प्रश्न
For a reaction : 
(i) Write the order and molecularity of this reaction.
(ii) Write the unit of k.
From the rate expression for the following reaction, determine the order of reaction and the dimension of the rate constant.
\[\ce{3NO_{(g)} -> N2O_{(g)}}\] Rate = k[NO]2
From the rate expression for the following reaction, determine the order of reaction and the dimension of the rate constant.
\[\ce{H2O2_{( aq)} + 3I^-_{( aq)} + 2H^+ -> 2H2O_{(l)} + I^-_3}\] Rate = k[H2O2][I−]
A reaction is first order in A and second order in B. How is the rate affected on increasing the concentration of B three times?
A reaction is first order in A and second order in B. How is the rate affected when the concentrations of both A and B are doubled?
Define the following term:
Pseudo first-order reaction
Rate of reaction for the combustion of propane is equal to:
\[\ce{C3H8_{(g)} + 5O2_{(g)} -> 3CO2_{(g)} + 4H2O_{(g)}}\]
What is the order of a reaction which has a rate expression; Rate = `"k"["A"]^(3/2)["B"]^1`?
Rate law for the reaction \[\ce{A + 2B -> C}\] is found to be Rate = k [A][B]. Concentration of reactant ‘B’ is doubled, keeping the concentration of ‘A’ constant, the value of rate constant will be ______.
Consider the reaction A ⇌ B. The concentration of both the reactants and the products varies exponentially with time. Which of the following figures correctly describes the change in concentration of reactants and products with time?
For a complex reaction:
(i) order of overall reaction is same as molecularity of the slowest step.
(ii) order of overall reaction is less than the molecularity of the slowest step.
(iii) order of overall reaction is greater than molecularity of the slowest step.
(iv) molecularity of the slowest step is never zero or non interger.
For a general reaction A → B, plot of concentration of A vs time is given in figure. Answer the following question on the basis of this graph.
(i) What is the order of the reaction?
(ii) What is the slope of the curve?
(iii) What are the units of rate constant?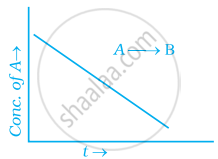
Why is the probability of reaction with molecularity higher than three very rare?
Why does the rate of any reaction generally decreases during the course of the reaction?
Why can’t molecularity of any reaction be equal to zero?
For a first order A → B, the reaction rate at reactant concentration of 0.01 m is found to be 2.0 × 10–5. The half-life period of reaction.
The number of molecules of the reactants taking part in a single step of the reaction is indicative of ______.
Identify the order of reaction from the following unit for its rate constant:
L mol–1 s–1
On heating compound (A) gives a gas (B) which is constituent of air. The gas when treated with H2 in the presence of catalyst gives another gas (C) which is basic in nature, (A) should not be ______.
Which of the following statement is true?
