Advertisements
Advertisements
प्रश्न
For a general reaction A → B, plot of concentration of A vs time is given in figure. Answer the following question on the basis of this graph.
(i) What is the order of the reaction?
(ii) What is the slope of the curve?
(iii) What are the units of rate constant?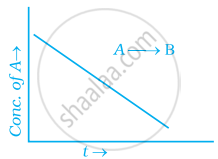
Advertisements
उत्तर
(i) Zero order reaction
(ii) [R] – [R]0 – kt
∴ Slope = – k
(iii) `(mol^-1L)/(s^-1)` = k[A]0 or k = mol–1 Ls–1
APPEARS IN
संबंधित प्रश्न
In a first order reaction x → y, 40% of the given sample of compound remains unreacted in 45 minutes. Calculate rate constant of the reaction.
A → B is a first order reaction with rate 6.6 × 10-5m-s-1. When [A] is 0.6m, rate constant of the reaction is
- 1.1 × 10-5s-1
- 1.1 × 10-4s-1
- 9 × 10-5s-1
- 9 × 10-4s-1
Write two factors that affect the rate of reaction.
The conversion of molecules X to Y follows second order kinetics. If concentration of X is increased to three times how will it affect the rate of formation of Y?
Mention the factors that affect the rate of a chemical reaction.
For a reaction R ---> P, half-life (t1/2) is observed to be independent of the initial concentration of reactants. What is the order of reaction?
The decomposition of N2O5(g) at 320K according to the following equation follows first order reaction:
`N_2O_(5(g))->2NO_(2(g))+1/2O_(2(g))`
The initial concentration of N2O5(g) is 1.24 x 10-2 mol. L-1 and after 60 minutes 0.20x10-2 molL-1. Calculate the rate constant of the reaction at 320K.
What is the order of a reaction which has a rate expression; Rate = `"k"["A"]^(3/2)["B"]^1`?
Molecularity of a reaction _____________.
For which type of reactions, order and molecularity have the same value?
Why can’t molecularity of any reaction be equal to zero?
Assertion: Rate constants determined from Arrhenius equation are fairly accurate for simple as well as complex molecules.
Reason: Reactant molecules undergo chemical change irrespective of their orientation during collision.
For a reaction \[\ce{Cl2l(g) + 2No(g) -> 2NaCl(g)}\] the rate law is expressed as rate= K[Cl2] [No]2 what is the order of the reaction?
For a first order A → B, the reaction rate at reactant concentration of 0.01 m is found to be 2.0 × 10–5. The half-life period of reaction.
For the reaction, \[\ce{A +2B → AB2}\], the order w.r.t. reactant A is 2 and w.r.t. reactant B. What will be change in rate of reaction if the concentration of A is doubled and B is halved?
The following data was obtained for chemical reaction given below at 975 K.
\[\ce{2NO(g) + 2H2(g) -> N2(g) + 2H2O(g)}\]
| [NO] | [H2] | Rate | |
| Mol L-1 | Mol L-1 | Mol L-1 s-1 | |
| (1) | 8 × 10-5 | 8 × 10-5 | 7 × 10-9 |
| (2) | 24 × 10-5 | 8 × 10-5 | 2.1 × 10-8 |
| (3) | 24 × 10-5 | 32 × 10-5 | 8.4 × 10-8 |
The order of the reaction with respect to NO is ______. (Integer answer)
For a chemical reaction starting with some initial concentration of reactant At as a function of time (t) is given by the equation,
`1/("A"_"t"^4) = 2 + 1.5 xx 10^-3` t
The rate of disappearance of [A] is ____ × 10-2 M/sec when [A] = 2 M.
[Given: [At] in M and t in sec.]
[Express your answer in terms of 10-2 M /s]
[Round off your answer if required]
Higher yield of NO in \[\ce{N2(g) + O2 <=> 2NO(g)}\] can be obtained at:
[ΔH of the reaction = +180.7 kJ mol−1]
- higher temperature
- lower temperature
- higher concentration of N2
- higher concentration of O2
Choose the correct answer from the options given below:
