Advertisements
Advertisements
प्रश्न
A sphere of radius 1.00 cm is placed in the path of a parallel beam of light of large aperture. The intensity of the light is 0.5 W cm−2. If the sphere completely absorbs the radiation falling on it, Show that the force on the sphere due to the light falling on it is the same even if the sphere is not perfectly absorbing.
Advertisements
उत्तर
Consider a sphere of centre O and radius OP. As shown in the figure, the radius OP of the sphere is making an angle θ with OZ. Let us rotate the radius about OZ to get another circle on the sphere. The part of the sphere between the circle is a ring of area `2pir^2sin θdθ`.
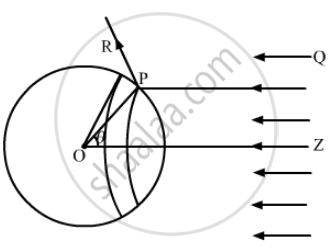
Consider a small part of area `ΔA` of the ring at point P.
Energy of the light falling on this part in time `Δt` ,
`ΔU = I Δ t (ΔA cos θ)`
As the light is reflected by the sphere along PR, the change in momentum ,
`Δp = 2 (ΔU)/c cos θ = 2/c I Δ t (ΔA cos^2 θ)`
Therefore , the force will be
`(Δp)/(Δt) = 2/c I ΔA cos^2 θ`
The Component of force on ΔA , along ZO , is
`(Δp)/(Δt) cos θ = 2/c I ΔA cos^3 θ`
Now , force action on the ring,
`dF = 2/c I (2pir^2 sin θ dθ) cos^3 θ`
The force on the entire sphere ,
`F = ∫_0^(pi/2) (4pir^2I)/c cos^3 θ sin θ dθ`
= `- ∫_0^(pi/2) (4pir^2I)/c cos^3 θd(cos θ)`
= `(pir^2I)/c`
APPEARS IN
संबंधित प्रश्न
Light of intensity 10−5 W m−2 falls on a sodium photo-cell of surface area 2 cm2. Assuming that the top 5 layers of sodium absorb the incident energy, estimate time required for photoelectric emission in the wave-picture of radiation. The work function for the metal is given to be about 2 eV. What is the implication of your answer?
Can we find the mass of a photon by the definition p = mv?
A hot body is placed in a closed room maintained at a lower temperature. Is the number of photons in the room increasing?
It is found that yellow light does not eject photoelectrons from a metal. Is it advisable to try with orange light or with green light?
The threshold wavelength of a metal is λ0. Light of wavelength slightly less than λ0 is incident on an insulated plate made of this metal. It is found that photoelectrons are emitted for some time and after that the emission stops. Explain.
Let nr and nb be the number of photons emitted by a red bulb and a blue bulb, respectively, of equal power in a given time.
The work function of a metal is hv0. Light of frequency v falls on this metal. Photoelectric effect will take place only if
Light of wavelength λ falls on a metal with work-function hc/λ0. Photoelectric effect will take place only if
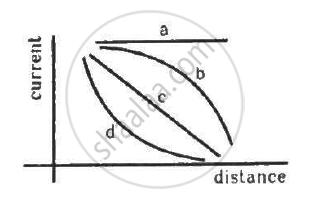
A point source causes photoelectric effect from a small metal plate. Which of the following curves may represent the saturation photocurrent as a function of the distance between the source and the metal?
An atom absorbs a photon of wavelength 500 nm and emits another photon of wavelength 700 nm. Find the net energy absorbed by the atom in the process.
(Use h = 6.63 × 10-34J-s = 4.14 × 10-15 eV-s, c = 3 × 108 m/s and me = 9.1 × 10-31kg)
A sphere of radius 1.00 cm is placed in the path of a parallel beam of light of large aperture. The intensity of the light is 0.5 W cm−2. If the sphere completely absorbs the radiation falling on it, find the force exerted by the light beam on the sphere.
(Use h = 6.63 × 10-34J-s = 4.14 × 10-15 eV-s, c = 3 × 108 m/s and me = 9.1 × 10-31kg)
The work function of a metal is 2.5 × 10−19 J. (a) Find the threshold frequency for photoelectric emission. (b) If the metal is exposed to a light beam of frequency 6.0 × 1014 Hz, what will be the stopping potential?
(Use h = 6.63 × 10-34J-s = 4.14 × 10-15 eV-s, c = 3 × 108 m/s and me = 9.1 × 10-31kg)
Define the terms "stopping potential' and 'threshold frequency' in relation to the photoelectric effect. How does one determine these physical quantities using Einstein's equation?
In photoelectric effect, the photoelectric current started to flow. This means that the frequency of incident radiations is ______.
Consider a metal exposed to light of wavelength 600 nm. The maximum energy of the electron doubles when light of wavelength 400 nm is used. Find the work function in eV.
Two monochromatic beams A and B of equal intensity I, hit a screen. The number of photons hitting the screen by beam A is twice that by beam B. Then what inference can you make about their frequencies?
Read the following paragraph and answer the questions.
| The figure shows the variation of photoelectric current measured in a photocell circuit as a function of the potential difference between the plates of the photocell when light beams A, B, C and D of different wavelengths are incident on the photocell. Examine the given figure and answer the following questions: |

- Which light beam has the highest frequency and why?
- Which light beam has the longest wavelength and why?
- Which light beam ejects photoelectrons with maximum momentum and why?
How would the stopping potential for a given photosensitive surface change if the intensity of incident radiation was decreased? Justify your answer.
A metallic plate exposed to white light emits electrons. For which of the following colours of light, the stopping potential will be maximum?
- Assertion (A): For the radiation of a frequency greater than the threshold frequency, the photoelectric current is proportional to the intensity of the radiation.
- Reason (R): Greater the number of energy quanta available, the greater the number of electrons absorbing the energy quanta and the greater the number of electrons coming out of the metal.
