Advertisements
Advertisements
प्रश्न
Balance the following chemical equation:

Advertisements
उत्तर
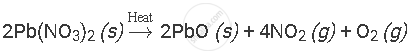
APPEARS IN
संबंधित प्रश्न
How will you indicate Endothermic reaction in a chemical equation?
Balance the given equation:
AI(OH)3  AI2O3 +H2O
AI2O3 +H2O
Potassium chlorate (KClO3) on heating forms potassium chloride and oxygen. Write a balanced equation for this reaction and indicate the evolution of gas.
Write a balanced chemical equation for the process of photosynthesis giving the physical states of all the substances involved and the conditions of the reaction.
Is Decomposition of calcium carbonate an endothermic reaction or an exothermic reaction?
Which of the following is not an endothermic reaction?
(a) CaCO3 → CaO + CO2
(b) 2H2O →2H2 + O2
(c) 6CO2 + 6H2O → C6H12O6 + 6O2
(d) C6H12O6 + 6O2 → 6CO2 + 6H2O
Why do we need to balance chemical equations?
What information do you get from the equation H2+ Cl2 → 2HCl ?
Write your observation and name the product when zinc reacts with dilute hydrochloric acid.
Dry raisins were soaked in water for 2 hours, to determine the percentage of water absorbed by raisins. Before final weighing of swollen raisins, the extra water left on the surface of soaked raisins was removed by:
The step(s) necessary for determining the percentage of water absorbed by raisins is/are:
Write the balanced chemical equation of the following reaction. iron + sulphuric acid → ferrous sulphate + hydrogen.
A chemical reaction is generally accompanied by certain external indications or characteristics. These include – change of – (a) colour (b) state (c) smell (d) evolution of gas (e) formation of precipitate (f) evolution or absorption of heat. With reference to change of colour – state the change in colour seen when the following are heated – mercury [II] oxide.
Name the following:
The silvery residue obtained on heating mercury [II] oxide.
Give an example of a chemical equation in which two reactants form:
three product
\[\ce{2KClO3->[MnO2] 2KCl + 3O2[g] - is a balanced equation.}\]
State why the compound MnO2 is written above the arrow.
Balance the following simple equation:
K + H2O → KOH + H2
Underline the compound in the equation given below, it is incorrectly balanced and write the correct balancing for the same.
C2H5OH + 3O2 → 2CO2 + 2H2O
Write scientific reason.
Adding zinc particles to a solution of copper sulphate makes the blue solution colorless.
Match the columns.
| Reactants | Products | Types of chemical reaction | ||
| 1. | MgH2 | → | Mg + H2 | Endothermic |
| 2. | 2H2S + SO2 | → | 3S + 2H2O | Oxidation |
| 3. | CaO + H2O | → | Ca(OH)2 + heat | Exothermic |
| Redox | ||||
