Advertisements
Advertisements
Question
Balance the following chemical equation:

Advertisements
Solution
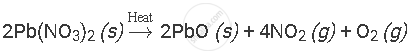
APPEARS IN
RELATED QUESTIONS
Write complete balanced equation for the following reaction:
Sulphur dioxide (gas) + Oxgyen (gas)  Sulphur trioxide (gas)
Sulphur trioxide (gas)
Write balanced chemical equation from the following information:
An aqueous calcium hydroxide solution (lime water) reacts with carbon dioxide gas to produce a solid calcium carbonate precipitate and water.
Explain, with example, how the physical states of the reactants and products can be shown in a chemical equation.
Carbon monoxide reacts with hydrogen under certain conditions to form methanol (CH3OH). Write a balanced chemical equation for this reaction indicating the physical states of reactants and product as well as the conditions under which this reaction takes place.
Name the type of chemical reaction shown by the following equation:
Fe + CuSO4 → FeSO4 +Cu
Write your observation for the following chemical reaction and name the product formed :
When dilute acetic acid is poured on baking soda.
Balance the following equation. Also name the product formed.
`"Zn" + "HCI" → "ZnCI" _2 + "H"_2`
Complete the following equation:
CH3COOH + NaOH →
To determine the percentage of water absorbed by raisins, raisins are soaked in water for:
Write the chemical equation for the following word equation and balance them.
Calcium oxide + Carbon dioxide → Calcium carbonate
What information does the following chemical equation convey?
Zn + H2SO4 → ZnSO4+ H2
Write the balanced chemical equation of the following reaction.
zinc sulphide + oxygen → zinc oxide + sulphur dioxide
Identify the type of reaction taking place of the following case and write the balanced chemical equation of the reaction:
Zinc reacts with silver nitrate to produce zinc nitrate and silver.
Write word equation for the following molecular equation:
FeSO4 + 2NaOH →Na2SO4 + Fe(OH)2 ↓
Word equation:
State why [↓] indication is seen after Fe(OH)2 and not after Na2SO4
Representation of the results of a chemical change – is a chemical equation.
For the equation: FeCl3 + 3NH4OH 3NH4Cl + Fe(OH)3 ↓
Answer the following:
Name the reactants and the products in the above equation.
An element X is trivalent. Write the balanced equation for the combustion of X in oxygen.
Write a balanced equation for the following word equation:
Aluminium + Oxygen → Aluminium oxide
Underline the compound in the equation given below, it is incorrectly balanced and write the correct balancing for the same.
6NaOH + 3Cl2 → 6NaCl + NaClO3 + 3H2O
Underline the compound in the equation given below, it is incorrectly balanced and write the correct balancing for the same.
C2H5OH + 3O2 → 2CO2 + 2H2O
Match the columns.
| Reactants | Products | Types of chemical reaction | ||
| 1. | MgH2 | → | Mg + H2 | Endothermic |
| 2. | 2H2S + SO2 | → | 3S + 2H2O | Oxidation |
| 3. | CaO + H2O | → | Ca(OH)2 + heat | Exothermic |
| Redox | ||||
