Advertisements
Advertisements
Question
Balance the following chemical equation:

Advertisements
Solution
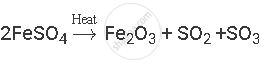
APPEARS IN
RELATED QUESTIONS
Translate the following statement into chemical equation and then balance it.
Potassium metal reacts with water to give potassium hydroxide and hydrogen gas.
State whether the following statement is true or false:
A chemical equation can be balanced easily by altering the formula of a reactant or product.
Correct and balance the following equation:
Ca + H2O  CaOH + H
CaOH + H
Write complete balanced equation for the following reaction:
Sulphur dioxide (gas) + Oxgyen (gas)  Sulphur trioxide (gas)
Sulphur trioxide (gas)
Write a balanced chemical equation for the process of photosynthesis giving the physical states of all the substances involved and the conditions of the reaction.
Give one example of a chemical reaction characterised by change in state.
Give one example of an endothermic reaction.
The chemical reaction between two substances is characterised by a change in colour from orange to green. These two substances are most likely to be:
(a) potassium dichromate solution and sulphur dioxide
(b) potassium permanganate solution and sulphur dioxide
(c) potassium permanganate solution and lemon juice
(d) potassium dichromate solution and carbon dioxide.
Explain the following reaction with one suitable example .
Decomposition reaction
Balance the following equation :
Pb3O4 → PbO + O2
Balance the following equation. Also name the product formed.
`"Na"+ "H"_2 "O" → "NaOH"+ "H"_2`
Explain the terms with examples.
Balanced equation
Write the answer to the following.
Explain the similarity and difference in two events, namely adding NaOH to water and adding CaO to water.
Balance the following equation:
C + H2SO4 → CO2 + H2O + SO2
Write word equation for the following molecular equation:
CaO + H20 → Ca (OH)2 + Δ
Word equation:
State why [+ Δ ] is not written in the reaction, if ZnO reacts with H2O.
Representation of the results of a chemical change – is a chemical equation.
For the equation: FeCl3 + 3NH4OH 3NH4Cl + Fe(OH)3 ↓
Answer the following:
What is the indications of the arrow between the reactants and the products and of the arrow pointing downwards at the end.
Balance the following simple equation:
H2 + Cl2 → HCl
With reference to a chemical equation state which of the statements 1 to 5 pertain to A or B.
A: Information provided by a chemical equation.
B: Limitations of a chemical equation.
- The nature of the individual elements.
- The speed of the reaction.
- The state of matter in which the substance is present.
- The completion of the reaction.
- The direction of the reaction.
What are products? Explain with the help of example.
The following reaction is an example of a `4"NH"_3("g") + "SO"_2 -> 4"NO"("g") + 6"H"_2"O"("g")`
- displacement reaction
- combination reaction
- redox reaction
- neutralisation reaction
