Advertisements
Advertisements
प्रश्न
Balance the following chemical equation:

Advertisements
उत्तर
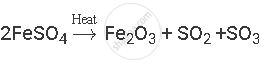
APPEARS IN
संबंधित प्रश्न
Methyl orange is used as an indicator. It shows colour change in acid, base and neutral substance. Tabulate your result as follows:
| Indicator | Colour change | Inference |
| Methyl Orange | No change | ...................... |
| .................... | Acid | |
| Yellow | ....................... |
Translate the following statement into chemical equation and then balance the equation:
Phosphorus burns in oxygen to give phosphorus pentoxide.
Translate the following statement into chemical equation and then balance the equation:
Carbon disulphide burns in air to give carbon dioxide and sulphur dioxide.
Give one example of a chemical reaction.
Balance the given equation:
H2O2  H2O + O2
H2O + O2
Balance the given equation:
NH3 + CuO  Cu +N2 +H2O
Cu +N2 +H2O
With the help of an appropriate example, justify that some of the chemical reactions are determined by Change in temperature.
Give chemical equation for the reaction involved in the above case.
State one characteristic of the chemical reaction which takes place when quicklime is treated with water
Write your observation for the following chemical reaction and name the product formed :
When manganese dioxide is added to potassium chlorate and heated.
Write symbolic representation for the following word equation and balance them :
Iron + Sulphur → Iron sulphide.
In the experiment for determining the percentage of water absorbed by raisins, we do the final weighing of the raisins after keeping them dipped in water for about one hour. For the accuracy of the result, the extra water from the surface of the soaked raisins is removed by
Write the chemical equation for the following word equation and balance them.
Nitrogen + Oxygen → Nitrogen monoxide
Write the balanced chemical equation of the following reaction.
sodium chloride + manganese dioxide + sulphuric acid → sodium hydrogen sulphate + manganese sulphate + water + chlorine.
Write word equation for the following molecular equation:
\[\ce{N2 + 3H2 ⇌[Fe - 450°C][200 atoms] 2NH3 + \triangle}\]
Word equation:
State why [+ Δ] indication is seen after NH3.
State what 200 atmospheres – indicates.
State the function of Fe in the above reaction.
In certain reaction a change of state is observed i.e. solid to liquid, liquid to gas etc. – State the change of state of the products – to give the respective reactant.
NH3 + HCl ⇌ NH4Cl
Representation of the results of a chemical change – is a chemical equation.
For the equation: FeCl3 + 3NH4OH 3NH4Cl + Fe(OH)3 ↓
Answer the following:
Name the reactants and the products in the above equation.
Give word equation for the following chemical reaction and give the names of the product formed.
ZnCO3 → ZnO + CO2
Write word equation for the following chemical reaction given below. Also state the observation seen in the case.
\[\ce{Na2SO3 + 2HCl ->[\triangle] 2NaCl + H2O + SO2}\]
Give an example of a chemical equation in which two reactants form:
three product
What is called thermolysis reactions?
