Advertisements
Advertisements
प्रश्न
Give one example of a chemical reaction.
Advertisements
उत्तर
Burning of magnesium ribbon in air to give white powder of magnesium oxide is an example of chemical reaction.
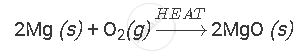
APPEARS IN
संबंधित प्रश्न
Correct and balance the following equation:
N + H  NH3
NH3
Balance the given equation:
NH3 + CuO  Cu +N2 +H2O
Cu +N2 +H2O
Aluminium burns in chlorine to form aluminium chloride (AlCl3). Write a balanced chemical equation for this reaction.
Write the balanced equation for the following reaction, and add the state symbols:
Sodium hydroxide reacts with sulphuric acid to produce sodium sulphate and water.
When metal X is treated with a dilute acid Y, then a gas Z is evolved which burns readily by making a little explosion.
(a) Name any two metals which can behave like metal X.
(b) Name any two acids which can behave like acid Y.
(c) Name the gas Z.
(d) Is the gas Z lighter than or heavier than air?
(e) Is the reaction between metal X and dilute acid Y, exothermic or endothermic?
(f) By taking a specific example of metal X and dilute acid Y, write a balanced chemical equation for the reaction which takes place. Also indicate physical states of all the reactants and products.
Balance the following chemical equation :
Na2CO3 + HCl → NaCI + H2O + CO2
Write symbolic representation for the following word equation and balance them :
Calcium oxide + Water → Calcium hydroxide
Dry raisins were soaked in water for 2 hours, to determine the percentage of water absorbed by raisins. Before final weighing of swollen raisins, the extra water left on the surface of soaked raisins was removed by:
The step(s) necessary for determining the percentage of water absorbed by raisins is/are:
Write the balanced chemical equation of the following reaction.
potassium dichromate + hydrochloric acid → Potassium chloride + chromium chloride + water + chlorine
(a) Give only one suitable chemical test to identify the following gases.
- Ammonia
- Sulphur dioxide
- Hydrogen Chloride
- Chlorine
- Carbon Dioxide
- Oxygen
- Hydrogen
(b) Select a basic gas mentioned in Q.1 (a). How is the basic nature suspected?
(c)Select acidic gases from the gases mentioned in Q.1 (a). How is the acidic nature suspected?
(d) State the gas responsible for bleaching action.
(e)Which gas turn blue cobalt chloride paper light pink?
When a potassium iodide solution is added to a solution of lead (II) nitrate in a test tube, a precipitate is formed.
What is the colour of this precipitate? Name the compound precipitated.
When a potassium iodide solution is added to a solution of lead (II) nitrate in a test tube, a precipitate is formed.
Write the balanced chemical equation for this reaction.
Name the following:
The gas evolved when a dilute acid is added to chalk [limestone].
Explain the term ‘chemical equation’.
What is meant by ‘reactants’ and ‘products’ in a chemical equation?
Underline the compound in the equation given below, it is incorrectly balanced and write the correct balancing for the same.
Na2SO3 + HCl → 2NaCl + H2O + SO2
Underline the compound in the equation given below, it is incorrectly balanced and write the correct balancing for the same.
6NaOH + 3Cl2 → 6NaCl + NaClO3 + 3H2O
Underline the compound in the equation given below, it is incorrectly balanced and write the correct balancing for the same.
3CuO + NH3 → 3Cu + 3H2O + N2
What is a product in a chemical equation?
