Advertisements
Advertisements
Question
Give one example of a chemical reaction.
Advertisements
Solution
Burning of magnesium ribbon in air to give white powder of magnesium oxide is an example of chemical reaction.
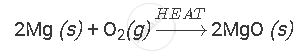
APPEARS IN
RELATED QUESTIONS
State whether the following statement is true or false:
A chemical equation can be balanced easily by altering the formula of a reactant or product.
What does the symbol (aq) represent in a chemical equation?
Balance the given equation:
H2O2  H2O + O2
H2O + O2
Giving examples, state the difference between balanced and unbalanced chemical equations.
Balance the given chemical equation:
NH3 → N2 + H2
State one characteristic of the chemical reaction which takes place when lemon juice is added gradually to potassium permanganate solution.
Which of the following does not involve a chemical reaction?
When water is added gradually to a white solid X, a hissing sound is heard and a lot of heat is produced forming a product Y. A suspension of Y in water is applied to the walls of a house during white washing. A clear solution of Y is also used for testing carbon dioxide gas in the laboratory.
(a) What could be solid X? Write its chemical formula.
(b) What could be product Y? Write its chemical formula.
(c) What is the common name of the solution of Y which is used for testing carbon dioxide gas?
(d) Write chemical equation of the reaction which takes place on adding water to slid X.
(e) Which characteristic of chemical reactions is illustrated by this example?
A white precipitate can be obtained by adding dilute sulphuric acid to:
(a) CuSO4 solution
(b) Nacl solution
(c) BaCl2 solution
(d) Na2SO4 solution
What is a chemical equation?
Write word equation for the following skeletal equation:
\[\ce{AgBr -> Ag + Br2}\]
A student weighed some raisins and recorded the weight as ‘x’. She then soaked the raisins in distilled water. After about 2 hours she removed the raisins, wiped them dry and weighed them again, and recorded that as ‘y’. The percentage of water absorbed by raisins may be determined using the relationship.
What is the limitation of the reaction given in question 2?
Balance the following equation:
Zn + KOH → K2ZnO2 + H2
Choose the correct alternative and rewrite the following sentence.
Which of the following is not required to find the pH of a given solution?
Name the following:
Two non-metal which react explosively when brought in close contact.
Balance the following simple equation:
KHCO3 → K2CO3 + H2O + CO2
Balance the following simple equation:
H2 + O2 → H2O
CaCO3 + 2HCl[dil.] → CaCl2 + H2O + CO2 [g]
State the information provided by the above chemical equation.
Reema took 5ml of Lead Nitrate solution in a beaker and added approximately 4ml of Potassium Iodide solution to it. What would she observe?
