Advertisements
Advertisements
प्रश्न
Balance the following chemical equation:

Advertisements
उत्तर
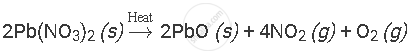
APPEARS IN
संबंधित प्रश्न
Balance the following chemical equation.
\[\ce{NaCl + AgNO3 -> AgCl + NaNO3}\]
Write the balanced chemical equation for the following reaction.
\[\ce{Calcium hydroxide + Carbon dioxide → Calcium carbonate + Water}\]
Find the odd man out:
Turmeric, Methyl Orange, Rose petals, Beetroot.
Balance the following equation:
Na + O2  Na2O
Na2O
Write any two observations in an activity which may suggest that a chemical reaction has taken place. Give an example in support of your answer.
Give one example of a chemical reaction characterised by formation of a precipitate.
Is Photosynthesis an endothermic reaction or an exothermic reaction?
Write your observations and name the product when
Iron nails are added to an aqueous solution of copper sulphate.
Dry raisins were soaked in water for 2 hours, to determine the percentage of water absorbed by raisins. Before final weighing of swollen raisins, the extra water left on the surface of soaked raisins was removed by:
Write the chemical equation for the following word equation and balance them.
Sodium reacts with water to form sodium hydroxide and hydrogen
Balance the following equation:
C + H2SO4 → CO2 + H2O + SO2
Write the balanced chemical equation of the following reaction. sodium hydroxide + sulphuric acid → sodium sulphate + water
Write the balanced chemical equation of the following reaction.
silver nitrate → silver + nitrogen dioxide + oxygen
\[\ce{MnO2 + 4HCl -> MnCl2 + 2H2O + Cl2}\]
0.02 moles of pure MnO2 is heated strongly with conc. HCl. Calculate the mass of acid required.
Write word equation for the following molecular equation:
\[\ce{CuSO4 + 2NaOH -> Na2SO4 + Cu(OH)2↓}\]
Word equation:
State the colour of the products.
Give a balanced equation for addition of iron to copper [II] sulphate solution. State the change in colour seen.
Write a balanced equation for the following word equation:
Calcium hydroxide + Ammonium chloride → Calcium chloride + Water + Ammonia
With reference to a chemical equation state which of the statements 1 to 5 pertain to A or B.
A: Information provided by a chemical equation.
B: Limitations of a chemical equation.
- The nature of the individual elements.
- The speed of the reaction.
- The state of matter in which the substance is present.
- The completion of the reaction.
- The direction of the reaction.
Which among the following statement(s) is(are) true? ‘Exposure of silver chloride to sunlight for a long duration turns grey due to
- The formation of silver by the decomposition of silver chloride.
- sublimation of silver chloride.
- decomposition of chlorine gas from silver chloride.
- oxidation of silver chloride.
Complete the following blank in the equation as indicated.
\[\ce{CaH2_{(s)} + 2H2O_{( aq)}-> Ca(OH)2_{(s)} + 2H2_{(g)}}\]
Grams: 42 g + ______ `→` ______ + ______
