Advertisements
Advertisements
Value of standard electrode potential for the oxidation of \[\ce{Cl-}\] ions is more positive than that of water, even then in the electrolysis of aqueous sodium chloride, why is \[\ce{Cl-}\] oxidised at anode instead of water?
Concept: undefined >> undefined
Which reference electrode is used to measure the electrode potential of other electrodes?
Concept: undefined >> undefined
Advertisements
Assertion: Cu is less reactive than hydrogen.
Reason: `E_((Cu^(2+))/(Cu))^Θ` is negative.
Concept: undefined >> undefined
Consider the figure and answer the following question.
If cell ‘A’ has ECell = 0.5V and cell ‘B’ has ECell = 1.1V then what will be the reactions at anode and cathode?
Concept: undefined >> undefined
At high pressure the following reaction is zero order.
\[\ce{2NH3(g) ->[1130 K][Platinum catalyst] N2(g) + 3H2(g)}\]
Which of the following options are correct for this reaction?
(i) Rate of reaction = Rate constant.
(ii) Rate of the reaction depends on concentration of ammonia.
(iii) Rate of decomposition of ammonia will remain constant until ammonia disappears completely.
(iv) Further increase in pressure will change the rate of reaction.
Concept: undefined >> undefined
Which of the following graphs is correct for a zero order reaction?
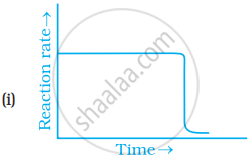

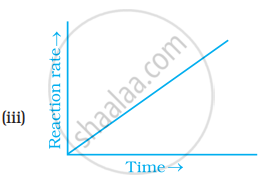

Concept: undefined >> undefined
Write the rate equation for the reaction `2A + B -> C` if the order of the reaction is zero.
Concept: undefined >> undefined
Derive an expression to calculate time required for completion of zero order reaction.
Concept: undefined >> undefined
For a zero order reaction will the molecularity be equal to zero? Explain.
Concept: undefined >> undefined
Arrange the following diagrams in correct sequence of steps involved in the mechanism of catalysis, in accordance with modern adsorption theory.

(a)

(b)

(c)

(d)

(e)
Concept: undefined >> undefined
In qualitative analysis when \[\ce{H2S}\] is passed through an aqueous solution of salt acidified with dil. \[\ce{HCl}\], a black precipitate is obtained. On boiling the precipitate with dil. \[\ce{HNO3}\], it forms a solution of blue colour. Addition of excess of aqueous solution of ammonia to this solution gives ______.
Concept: undefined >> undefined
A brown ring is formed in the ring test for \[\ce{NO3^{-}}\] ion. It is due to the formation of ______.
Concept: undefined >> undefined
In the preparation of HNO3, we get NO gas by catalytic oxidation of ammonia. The moles of NO produced by the oxidation of two moles of NH3 will be ______.
Concept: undefined >> undefined
\[\ce{PCl5}\] reacts with finely divided silver on heating and a white silver salt is obtained, which dissolves on adding excess aqueous \[\ce{NH3}\] solution. Write the reactions involved to explain what happens.
Concept: undefined >> undefined
The electronic configuration of \[\ce{Cu(II)}\] is 3d9 whereas that of \[\ce{Cu(I)}\] is 3d10. Which of the following is correct?
Concept: undefined >> undefined
Which of the following complexes formed by \[\ce{Cu^2+}\] ions is most stable?
Concept: undefined >> undefined
When 0.1 mol \[\ce{CoCl3 (NH3)5}\] is treated with excess of \[\ce{AgNO3}\], 0.2 mol of \[\ce{AgCl}\] are obtained. The conductivity of solution will correspond to ______.
Concept: undefined >> undefined
When 1 mol \[\ce{CrCl3.6H2O}\] is treated with excess of \[\ce{AgNO3}\], 3 mol of \[\ce{AgCl}\] are obtained. The formula of the complex is ______.
Concept: undefined >> undefined
The correct \[\ce{IUPAC}\] name of \[\ce{[Pt(NH3)2Cl2]}\] is ______.
Concept: undefined >> undefined
The stabilisation of coordination compounds due to chelation is called the chelate effect. Which of the following is the most stable complex species?
Concept: undefined >> undefined
