Advertisements
Advertisements
प्रश्न
When 0.1 mol \[\ce{CoCl3 (NH3)5}\] is treated with excess of \[\ce{AgNO3}\], 0.2 mol of \[\ce{AgCl}\] are obtained. The conductivity of solution will correspond to ______.
विकल्प
1:3 electrolyte
1:2 electrolyte
1:1 electrolyte
3:1 electrolyte
Advertisements
उत्तर
When 0.1 mol \[\ce{CoCl3 (NH3)5}\] is treated with excess of \[\ce{AgNO3}\], 0.2 mol of \[\ce{AgCl}\] are obtained. The conductivity of solution will correspond to 1:2 electrolyte.
Explanation:
One mole of \[\ce{AgNO3}\] precipitates one mole of chloride ion. In the above reaction, when 0.1 mole \[\ce{CoCl3 (NH3)5}\] is treated with excess of \[\ce{AgNO3}\], 0.2 mol of \[\ce{AgCl}\] are obtained thus, there must be two free chloride ions in the solution of electrolyte.
So, molecular formula of complex will be \[\ce{[CO(NH3)5Cl]Cl2}\] and electrolytic solution must contain \[\ce{[CO(NH3)5Cl]^{2+}}\] and two CT as constituent ions. Thus, it is 1:2 electrolyte.
\[\ce{[Co(NH3)5Cl]Cl2 -> [Co(NH3)5Cl]^{2+} (aq) + 2Cl^{-} (aq)}\]
APPEARS IN
संबंधित प्रश्न
Write IUPAC names of the following compounds:

Write structures of compounds A, B and C in of the following reactions

Write the IUPAC name of the following complex:
[Co(NH3)5(CO3)]Cl
Write the structures of compounds A, B and C in the following reactions
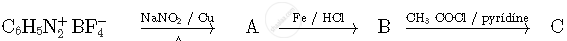
Write structures of compounds A and B of the following reaction :

Which of the following is non-ionizable?
Which of the following complexes formed by \[\ce{Cu^2+}\] ions is most stable?
A coordination compound \[\ce{CrCl3.4H2O}\] precipitates silver chloride when treated with silver nitrate. The molar conductance of its solution corresponds to a total of two ions. Write structural formula of the compound and name it.
What is the relationship between observed colour of the complex and the wavelength of light absorbed by the complex?
In which of the following compounds, the oxidation number of iodine is fractional?
The oxidation state of Fe in the brown ring complex [F3(H2O)5NO]SO4 is
The oxidation number d-arbitral occupation and co-ordination number of Cr in the complex cis [Cr(en)2Cl2]Cl are respectively.
Which of the following ligands can exhibit linkage isomerism?
What are Homoleptic complexes?
Given below are two statements.
Statement I: In CuSO4·5H2O, Cu-O bonds are present.
Statement II: In CuSO4·5H2O, ligands coordinating with Cu(II) ion are O- and S-based ligands.
In light of the above statement, choose the correct answer from the options given below.
What is a chelate complex?
Does ionization isomer for the following compound exist? Justify your answer.
\[\ce{Hg[Co(SCN)4]}\]
What is meant by the chelate effect? Give an example.
Explain the following, giving two examples:
Heteroleptic
Give two examples of didentate ligands.
