Definitions [1]
Organic compounds containing carbon-oxygen double bond, i.e. \[\mathrm{>C=O}\] group, are known as carbonyl compounds.
Key Points
- Carbonyl group: The ≻C=O group (carbonyl carbon + carbonyl oxygen) — a key functional group in organic chemistry.
- Carbonyl compounds: Aldehydes and ketones, both containing ≻C=O as their functional group.
- Aldehydes: –CHO (formyl group); carbonyl C bonded to at least one H.
- Ketones: ≻C=O (ketonic carbonyl group); carbonyl C bonded to two alkyl/aryl groups (R=R′ or R≠R′).
- Carboxylic acids: –COOH (carboxyl group); –OH attached to ≻C=O makes them distinct from aldehydes/ketones.
Preparation of aliphatic aldehydes and ketones
By oxidation of alcohols:
\[\ \begin{array}{r@{\;}c@{\;}l} \mathrm{R} & & \\ & \backslash & \\ & & \mathrm{CH}-\mathrm{OH} \\ & / & \\ \mathrm{R}' & & \end{array} + [\mathrm{O}] \xrightarrow[\text{Or KMnO}_4]{\text{K}_2\text{Cr}_2\text{O}_7/\text{H}_2\text{SO}_4} \begin{array}{r@{\;}c@{\;}l} \mathrm{R} & & \\ & \backslash & \\ & & \mathrm{C}=\mathrm{O} \\ & / & \\ \mathrm{R}' & & \end{array} + \mathrm{H}_2\mathrm{O}\]
When,
- R' = H then 1° alcohol to aldehyde.
- R' = alkyl group then 2º alcohol to ketone.
By dehydrogenation of alcohols:
\[ \begin{array}{r@{\;}c@{\;}l} \mathrm{R} & & \\ & \backslash & \\ & & \mathrm{CH}-\mathrm{OH} \\ & / & \\ \mathrm{R}' & & \end{array} \xrightarrow[\text{573 K}]{\text{Cu}} \begin{array}{r@{\;}c@{\;}l} \mathrm{R} & & \\ & \backslash & \\ & & \mathrm{C}=\mathrm{O} \\ & / & \\ \mathrm{R}' & & \end{array} + \mathrm{H}_2 \]
When
- R' = H then 1° alcohol to aldehyde.
- R' = alkyl group the 2° alcohol to ketone.
By acid chloride:
\[ \mathrm{R} - \overset{\displaystyle \mathrm{O}}{\overset{||}{\mathrm{C}}} - \mathrm{Cl} + \mathrm{H}_2 \xrightarrow[\text{Rosenmund Reduction}]{\text{Pd}-\text{BaSO}_4} \underset{\text{Aldehyde}}{\mathrm{R} - \overset{\displaystyle \mathrm{O}}{\overset{||}{\mathrm{C}}} - \mathrm{H}} + \mathrm{HCl} \]
2RMgX + CdCl2 → R2Cd + 2MgXCl
\[\ 2\mathrm{R}' - \overset{\displaystyle \mathrm{O}}{\overset{||}{\mathrm{C}}} - \mathrm{Cl} + \mathrm{R}_2\mathrm{Cd} \longrightarrow 2\mathrm{R}' - \underset{\text{Ketone}}{\overset{\displaystyle \mathrm{O}}{\overset{||}{\mathrm{C}}}} - \mathrm{R} + \mathrm{CdCl}_2 \]
From nitriles and esters:
\[ \mathrm{R} - \mathrm{CN} \xrightarrow[\text{(ii) }\mathrm{H}_2\mathrm{O}]{\text{(i) }\mathrm{AlH}(i\text{Bu})_2} \underset{\text{Aldehyde}}{\mathrm{R} - \mathrm{CHO}} \]
\[ \mathrm{CH}_3(\mathrm{CH}_2)_9 - \overset{\displaystyle \mathrm{O}}{\overset{||}{\mathrm{C}}} - \mathrm{OC}_2\mathrm{H}_5 \xrightarrow[\text{(ii) }\mathrm{H}_2\mathrm{O}]{\text{(i) DIBAL-H}} \mathrm{CH}_3(\mathrm{CH}_2)_9 - \underset{\text{Aldehyde}}{\overset{\displaystyle \mathrm{O}}{\overset{||}{\mathrm{C}}} - \mathrm{H}} \]
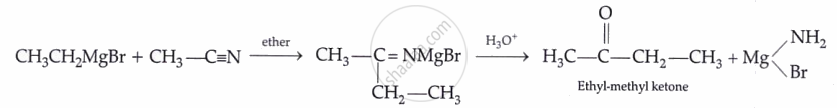 From hydrocarbons
From hydrocarbons
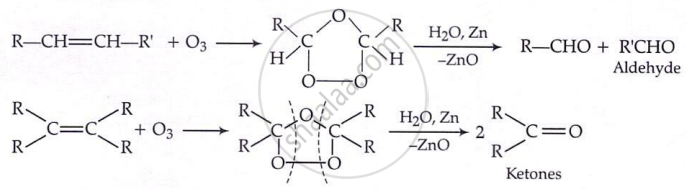
By ozonolysis:
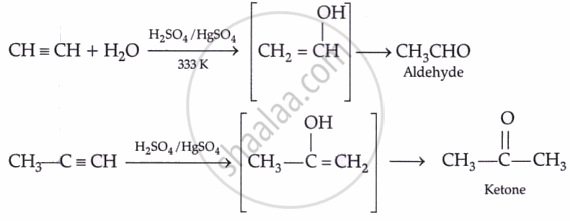
By hydration:
- Most aldehydes are liquids (except HCHO = gas); ketones of lower order are colourless liquids with a pleasant odour.
- Higher BP than corresponding hydrocarbons but lower than alcohols (no H-bonding between molecules, but dipole-dipole interactions).
- Lower members are soluble in water (H-bonding with water); higher members are insoluble (large alkyl groups).
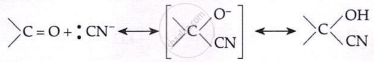
Addition of HCN:
\[\text{HCN} + \text{OH}^- \rightleftharpoons :\text{CN}^- + \text{H}_2\text{O}\]
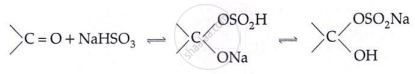
Addition of NaHSO3:
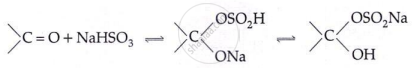
Addition of Grignard reagent:
Addition of alcohols:
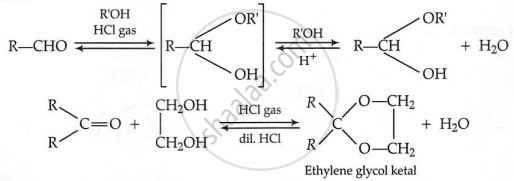
- Formaldehyde: Used in making Bakelite (phenol-formaldehyde resin), as a preservative (formalin = 40% HCHO).
- Acetaldehyde: Used in the preparation of acetic acid and ethanol.
- Acetone: Solvent (nail polish remover), used in the manufacture of chloroform.
- Benzaldehyde: Used in perfumes and dyes.
Important Questions [42]
- Write Two Uses of Formaldehyde
- How Are Ketones Classified?
- Give Iupac Name
- Write chemical reactions for the following conversions: Ethyl bromide to ethyl methyl ether.
- Compound Having General Formula
- Write the Structures and IUPAC Names of the α - Methyl Butyraldehyde.
- Explain the Structure of Carbonyl Functional Group
- Write chemical equation for the following :Rosenmund reduction
- How Will You Convert Calcium Acetate to Acetaldehyde?
- What is the action of phenyl hydrazine on propanone?
- What is the action of Zn – Hg / conc. HCl on propanone?
- What is the action of Sodium bisulphite on propanone?
- Ozonolysis of 2, 3 dimethyl but-2-ene, followed by decomposition by Zn dust and water gives ______.
- Write Only Reactions for the Preparation of Benzophenone from Benzonitrile.
- Write a reaction for the following conversion. Benzene to Benzaldehyde.
- Write chemical equation for the following:Gatterman - Koch formylation
- Write a Note on ‘Aldol Condensation’.
- Write preparation of propanone by using ethanoyl chloride and dimethyl cadmium.
- How Will You Convert Benzoic Acid to M-bromobenzoic Acid?
- Acetaldehyde, when treated with which among the following reagents does NOT undergo addition reaction?
- Write balanced chemical equations for action of ammonia on - formaldehyde
- How Are the Following Compounds Prepared? benzaldehyde from benzene.
- Explain the Mechanism of Alkaline Hydrolysis of Tert-butyl Bromide with Energy Profile Diagram.
- How Are the Following Compounds Prepared? Acetophenone from Benzene
- How Are the Following Compounds Prepared? Benzaldehyde from Benzoyl Chloride
- Write Balanced Chemical Equations for Action of Ammonia on - Acetaldehyde
- Write Balanced Chemical Equations for Action of Ammonia on - Acetone
- What is the Action of the Following Reagents on Ethanoic Acid?
- Write the Structure of Phenylmethanamine.
- What Are Amines?
- How will you convert sodium acetate to methane?
- Write the name of product formed, when acetone is treated with 2, 4-dinitrophenyl hydrazine.
- What is the action of sodium hypoiodite on acetone?
- The product of following reaction is CHA3−CH=CH−CHA2−CHO→ii) HA3OA+i) LiAlHA4 ______?
- The product of the following reaction is O.........||.........CA2HA5−C−CHA3→ΔHA2/Ni..?
- How is 4-methylpent-3en-2-one Obtained from Propan-2-one?
- Write Mechanism of Aldol Addition Reaction.
- Formalin is 40% Aqueous Solution of
- Write a Note on Self Oxidation-reduction Reaction of Aldehyde with Suitable Example.
- How Will You Convert Acetone to Acetone Cyanohydrin?
- Explain the following reaction: Cannizzaro reaction
- Write the chemical reaction involved in Cannizzaro reaction of methanal.
Concepts [9]
- Concepts of Aldehydes, Ketones, and Carboxylic Acids
- Nomenclature of Aldehydes and Ketones
- Nature of Carbonyl Group
- Preparation of Aldehydes and Ketones
- Physical Properties of Aldehydes and Ketones
- Chemical Reactions of Aldehydes and Ketones - Nucleophilic Addition Reactions
- Reactivity of Alpha Hydrogen in Aldehydes
- Uses of Aldehydes and Ketones
- Chemical Reactions of Aldehydes and Ketones - Other Reactions
