Advertisements
Advertisements
प्रश्न
Suggest reasons why the B–F bond lengths in BF3 (130 pm) and `"BF"_4^(-)` (143 pm) differ.
Advertisements
उत्तर
The B–F bond length in BF3 is shorter than the B–F bond length `"BF"_4^(-)`. BF3 is an electron-deficient species. With a vacant p-orbital on boron, the fluorine and boron atoms undergo pπ – pπ back-bonding to remove this deficiency. This imparts a double-bond character to the B–F bond.

This double-bond character causes the bond length to shorten in BF3 (130 pm). However, when BF3 coordinates with the fluoride ion, a change in hybridisation from sp2 (in BF3) to sp3 (in `"BF"_4^(-)` ) occurs. Boron now forms 4σ bonds and the double-bond character is lost. This accounts for a B–F bond length of 143 pm in `"BF"_4^(-)` ion.

संबंधित प्रश्न
How would you explain the lower atomic radius of Ga as compared to Al?
What do you understand by inert pair effect?
Write a balanced equation for Al + NaOH → ?
Write a balanced equation for B2H6 + NH3 → ?
Which of the following oxides is acidic in nature?
The exhibition of highest co-ordination number depends on the availability of vacant orbitals in the central atom. Which of the following elements is not likely to act as central atom in \[\ce{MF^{3-}6}\]?
A compound X, of boron reacts with NH3 on heating to give another compound Y which is called inorganic benzene. The compound X can be prepared by treating BF3 with Lithium aluminium hydride. The compounds X and Y are represented by the formulas.
Dry ice is ______.
Which of the following statements are correct. Answer on the basis of Figure.
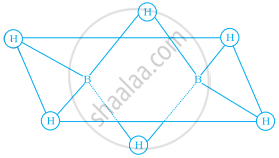
(i) The two birdged hydrogen atoms and the two boron atoms lie in one plane;
(ii) Out of six B – H bonds two bonds can be described in terms of 3 centre 2-electron bonds.
(iii) Out of six B – H bonds four B – H bonds can be described in terms of 3 centre 2 electron bonds;
(iv) The four-terminal B – H bonds are two centre-two electron regular bonds.
Aluminium dissolves in mineral acids and aqueous alkalies and thus shows amphoteric character. A piece of aluminium foil is treated with dilute hydrochloric acid or dilute sodium hydroxide solution in a test tube and on bringing a burning matchstick near the mouth of the test tube, a pop sound indicates the evolution of hydrogen gas. The same activity when performed with concentrated nitric acid, reaction doesn’t proceed. Explain the reason.
Explain the following:
PbX2 is more stable than PbX4.
Match the species given in Column I with properties given in Column II.
| Column I | Column II |
| (i) Diborane | (a) Used as a flux for soldering metals |
| (ii) Galluim | (b) Crystalline form of silica |
| (iii) Borax | (c) Banana bonds |
| (iv) Aluminosilicate | (d) Low melting, high boiling, useful for measuring high temperatures |
| (v) Quartz | (e) Used as catalyst in petrochemical industries |
Match the species given in Column I with the hybridisation given in Column II.
| Column I | Column II |
| (i) Boron in [B(OH)4]– | (a) sp2 |
| (ii) Aluminium in [Al(H2O)6]3+ | (b) sp3 |
| (iii) Boron in B2H6 | (c) sp3d2 |
| (iv) Carbon in Buckminsterfullerene | |
| (v) Silicon in \[\ce{SiO^{4-}4}\] | |
| (vi) Germanium in [GeCl6]2– |
Account for the following observations:
PbO2 is a stronger oxidising agent than SnO2
Account for the following observations:
The +1 oxidation state of thallium is more stable than its +3 state.
Three pairs of compounds are given below. Identify that compound in each of the pairs which has group 13 element in more stable oxidation state. Give reason for your choice. State the nature of bonding also.
TlCl3, TlCl
BCl3 exists as monomer whereas AlCl3 is dimerised through halogen bridging. Give reason. Explain the structure of the dimer of AlCl3 also.
Boron fluoride exists as BF3 but boron hydride doesn’t exist as BH3. Give reason. In which form does it exist? Explain its structure.
