Advertisements
Advertisements
प्रश्न
In some of the reactions thallium resembles aluminium, whereas in others it resembles with group I metals. Support this statement by giving some evidences.
Advertisements
उत्तर
Thallium belongs to group 13 of the periodic table. The most common oxidation state for this group is +3. However, heavier members of this group also display the +1 oxidation state. This happens because of the inert pair effect. Aluminium displays the +3 oxidation state and alkali metals display the +1 oxidation state. Thallium displays both the oxidation states. Therefore, it resembles both aluminium and alkali metals.
Thallium, like aluminium, forms compounds such as TlCl3 and Tl2O3. It resembles alkali metals in compounds Tl2O and TlCl.
संबंधित प्रश्न
How can you explain higher stability of BCl3 as compared to TlCl3?
Suggest reasons why the B–F bond lengths in BF3 (130 pm) and `"BF"_4^(-)` (143 pm) differ.
How would you explain the lower atomic radius of Ga as compared to Al?
Write a balanced equation for Al + NaOH → ?
Write a balanced equation for B2H6 + NH3 → ?
Which of the following oxides is acidic in nature?
The exhibition of highest co-ordination number depends on the availability of vacant orbitals in the central atom. Which of the following elements is not likely to act as central atom in \[\ce{MF^{3-}6}\]?
A compound X, of boron reacts with NH3 on heating to give another compound Y which is called inorganic benzene. The compound X can be prepared by treating BF3 with Lithium aluminium hydride. The compounds X and Y are represented by the formulas.
Which of the following statements are correct. Answer on the basis of Figure.
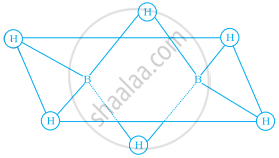
(i) The two birdged hydrogen atoms and the two boron atoms lie in one plane;
(ii) Out of six B – H bonds two bonds can be described in terms of 3 centre 2-electron bonds.
(iii) Out of six B – H bonds four B – H bonds can be described in terms of 3 centre 2 electron bonds;
(iv) The four-terminal B – H bonds are two centre-two electron regular bonds.
Explain why the following compounds behave as Lewis acids?
AlCl3
Explain the following:
PbX2 is more stable than PbX4.
Explain the following:
Electron gain enthalpy of chlorine is more negative as compared to fluorine.
Identify the compounds A, X and Z in the following reactions:
\[\ce{A + 2HCl + 5H2O -> 2NaCl + X}\]
Identify the compounds A, X and Z in the following reactions:
\[\ce{X ->[Δ][370 K] HBO2 ->[Δ][> 370 K] Z}\]
Account for the following observations:
Though fluorine is more electronegative than chlorine yet BF3 is a weaker Lewis acid than BCl3
Account for the following observations:
The +1 oxidation state of thallium is more stable than its +3 state.
A nonmetallic element of group 13, used in making bullet proof vests is extremely hard solid of black colour. It can exist in many allotropic forms and has unusually high melting point. Its trifluoride acts as Lewis acid towards ammonia. The element exihibits maximum covalency of four. Identify the element and write the reaction of its trifluoride with ammonia. Explain why does the trifluoride act as a Lewis acid.
Boron compounds behave as Lewis acids because of their ______.
