Advertisements
Advertisements
प्रश्न
A compound X, of boron reacts with NH3 on heating to give another compound Y which is called inorganic benzene. The compound X can be prepared by treating BF3 with Lithium aluminium hydride. The compounds X and Y are represented by the formulas.
विकल्प
B2H6, B3N3H6
B2O3, B3N3H6
BF3, B3N3H6
B3N3H6, B2H6
Advertisements
उत्तर
B2H6, B3N3H6
Explanation:
A compound X, of boron, reacts with NH3 on heating to give another compound YY which is called inorganic benzene.
\[\ce{\underset{X(Diborance)}{3H2H6} + 6NH3 -> 3[BH2(NH3)2]+ [BH4]- ->[heat] \underset{Y(Borazole Inorganic Benzene)}{2B3N3H6} + 12H2}\]
\[\ce{4BF3 + 3LiAlH4 -> \underset{X}{2B2H6} + 3LiF + 3AlF3}\]
APPEARS IN
संबंधित प्रश्न
In some of the reactions thallium resembles aluminium, whereas in others it resembles with group I metals. Support this statement by giving some evidences.
What do you understand by inert pair effect?
Write a balanced equation for B2H6 + NH3 → ?
The geometry of a complex species can be understood from the knowledge of type of hybridisation of orbitals of central atom. The hybridisation of orbitals of central atom in [Be(OH)4]– and the geometry of the complex are respectively.
The exhibition of highest co-ordination number depends on the availability of vacant orbitals in the central atom. Which of the following elements is not likely to act as central atom in \[\ce{MF^{3-}6}\]?
Which of the following statements are correct. Answer on the basis of Figure.
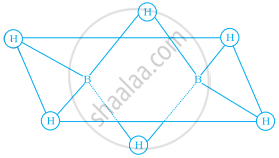
(i) The two birdged hydrogen atoms and the two boron atoms lie in one plane;
(ii) Out of six B – H bonds two bonds can be described in terms of 3 centre 2-electron bonds.
(iii) Out of six B – H bonds four B – H bonds can be described in terms of 3 centre 2 electron bonds;
(iv) The four-terminal B – H bonds are two centre-two electron regular bonds.
Explain why the following compounds behave as Lewis acids?
AlCl3
When BCl3 is treated with water, it hydrolyses and forms [B[OH]4]– only whereas AlCl3 in acidified aqueous solution forms [Al(H2O)6]3+ ion. Explain what is the hybridisation of boron and aluminium in these species?
Explain the following:
Boron does not exist as B3+ ion.
Explain the following:
Pb4+ acts as an oxidising agent but Sn2+ acts as a reducing agent.
Explain the following:
Electron gain enthalpy of chlorine is more negative as compared to fluorine.
Identify the compounds A, X and Z in the following reactions:
\[\ce{A + 2HCl + 5H2O -> 2NaCl + X}\]
Describe the general trends in the following properties of the elements in Groups 13 and 14.
Metallic character
Account for the following observations:
Though fluorine is more electronegative than chlorine yet BF3 is a weaker Lewis acid than BCl3
Three pairs of compounds are given below. Identify that compound in each of the pairs which has group 13 element in more stable oxidation state. Give reason for your choice. State the nature of bonding also.
TlCl3, TlCl
Three pairs of compounds are given below. Identify that compound in each of the pairs which has group 13 element in more stable oxidation state. Give reason for your choice. State the nature of bonding also.
InCl3, InCl
A group 13 element ‘X’ reacts with chlorine gas to produce a compound XCl3. XCl3 is electron deficient and easily reacts with NH3 to form \[\ce{Cl3X –> NH3}\] adduct; however, XCl3 does not dimerize X is ______.
Which one of the following is the correct statement?
