Advertisements
Advertisements
प्रश्न
How are the following compounds prepared?
benzaldehyde from benzoyl chloride
Advertisements
उत्तर
Benzaldehyde from benzoyl chloride

Rosenmund Reduction
APPEARS IN
संबंधित प्रश्न
Predict the products of the following reactions:

Predict the product of the following reaction:

What is meant by the following term? Give an example of the reaction in the following case.
Acetal
What is meant by the following term? Give an example of the reaction in the following case.
Semicarbazone
What is meant by the following term? Give an example of the reaction in the following case.
Oxime
What is meant by the following term? Give an example of the reaction in the following case.
Schiff’s base
Complete the synthesis by giving missing starting material, reagent or product.

Write the main product formed when propanal reacts with the following reagents:
2 moles of 3 CH OH in presence of dry HCl
Benzaldehyde can be obtained from benzal chloride. Write reactions for obtaining benzalchloride and then benzaldehyde from it.
Alkenes 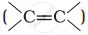 and carbonyl compounds
and carbonyl compounds 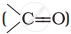 , both contain a π bond but alkenes show electrophilic addition reactions whereas carbonyl compounds show nucleophilic addition reactions. Explain.
, both contain a π bond but alkenes show electrophilic addition reactions whereas carbonyl compounds show nucleophilic addition reactions. Explain.
Carboxylic acids contain carbonyl group but do not show the nucleophilic addition reaction like aldehydes or ketones. Why?
Identify the compounds A, B and C in the following reaction.
\[\ce{CH3 - Br ->[Mg/ether] (A) ->[(i) CO][(ii) Water] (B) ->[CH3OH/H+][Δ] (C)}\]
A Idol condensation will not be observed in
Write the name of product formed, when acetone is treated with 2, 4-dinitrophenyl hydrazine.
What happens when propanone is treated with CH3MgBr and then hydrolysed?
What happens when ethanal is treated with excess ethanol and acid?
Which of the following is most reactive in nucleophilic addition reactions?
Aldehydes and ketones react with hydroxylamine to form ______.
Draw structures of the given derivatives.
The ethylene ketal of hexan-3-one
Draw the structure of the following derivative.
The ethylene ketal of hexane-3-one
Draw structure of the following derivative.
The ethylene ketal of hexane-3-one
Draw structures of the following derivative.
The ethylene ketal of hexan-3-one
Draw the structure of the following derivative.
Acetaldehydedimethylacetal
Draw structure of the following derivative.
The ethylene ketal of hexan-3-one
Draw structures of the following derivatives.
Acetaldehydedimethylacetal
Draw the structure of the following derivative.
Acetaldehydedimethylacetal
Complete the following reaction:
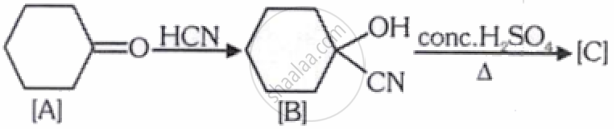
[C] is __________.
