Advertisements
Advertisements
प्रश्न
Alkenes 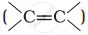 and carbonyl compounds
and carbonyl compounds 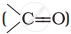 , both contain a π bond but alkenes show electrophilic addition reactions whereas carbonyl compounds show nucleophilic addition reactions. Explain.
, both contain a π bond but alkenes show electrophilic addition reactions whereas carbonyl compounds show nucleophilic addition reactions. Explain.
Advertisements
उत्तर
Carbonyl group is polar in nature. Due to larger electronegativity of oxygen as compared to carbon, carbon acquires partial positive charge while O acquires partial negative charge.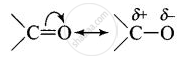
Because of slight positive charge on C atom, it is attacked by nucleophiles and, therefore, undergoes nucleophilic addition reaction.
Ethylenic double bond is a non-polar bond and is a source of electrons. Therefore, it is attacked by electrophiles and undergoes electrophilic addition reactions.
APPEARS IN
संबंधित प्रश्न
Acetaldehyde, when treated with which among the following reagents does NOT undergo addition reaction?
(A) Ammonia
(B) Hydroxylamine
(C) Ammoniacal silver nitrate
(D) Semicarbazide
Write balanced chemical equations for action of ammonia on - formaldehyde
Draw the structure of the semicarbazone of ethanal.
Write the products formed when CH3CHO reacts with the following reagents : HCN
Write the products formed when CH3CHO reacts with the following reagents : H2N – OH
Predict the product of the following reaction:

What is meant by the following term? Give an example of the reaction in the following case.
Acetal
What is meant by the following term? Give an example of the reaction in the following case.
Ketal
Predict the products formed when cyclohexanecarbaldehyde reacts with the following reagents.
PhMgBr and then H3O+
What is the action of the following reagents on ethanoic acid?
1) `LiAlH_4"/"H_3O^+`
2) `PCl_3 , "heat"`
3) `P_2O_5, "heat"`
Write the structure of Phenylmethanamine.
How will you convert sodium acetate to methane?
Write the main product formed when propanal reacts with the following reagents:
2 moles of 3 CH OH in presence of dry HCl
Grignard reagent on reaction with acetone forms.
The product of following reaction is
\[\ce{CH3 - CH = CH - CH2 - CHO ->[i) LiAlH4][ii) H3O+]}\] ______?
Which of the following is most reactive in nucleophilic addition reactions?
Draw structures of the following derivative.
The ethylene ketal of hexan-3-one
Which of the following will not be formed when calcium formate is distilled with calcium acetate?
