Advertisements
Advertisements
प्रश्न
Compounds A and C in the following reaction are:
\[\ce{CH3CHO ->[(i) CH3MgBr][(ii) H2O] (A) ->[H2SO4, Δ] (B) ->[Hydroboration oxidation] (C)}\]
विकल्प
identical
positional isomers
functional isomers
optical isomers
Advertisements
उत्तर
positional isomers
Explanation:
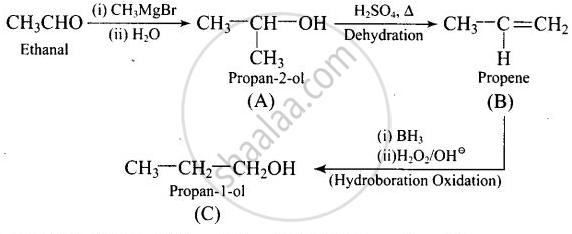
Thus, \[\begin{array}{cc}
\ce{CH3CH - OH}\\
|\phantom{....}\\
\ce{CH3}\phantom{..}
\end{array}\] and \[\ce{CH3 - CH2 - CH2OH}\] are positional isomers.
APPEARS IN
संबंधित प्रश्न
A compound 'A' of molecular formula C2H3OCl undergoes a series of reactions as shown below. Write the structures of A, B, C and D in the following reactions :

Write the products formed when CH3CHO reacts with the following reagents: CH3CHO in the presence of dilute NaOH
What is meant by the following term? Give an example of the reaction in the following case.
Aldol
Write structural formulas and names of four possible aldol condensation products from propanal and butanal. In each case, indicate which aldehyde acts as nucleophile and which as electrophile.
Describe the following:
Cross aldol condensation
Give reasons Acetylation of aniline reduces its activation effect.
Why is alpha (α) hydrogen of carbonyl compounds acidic in nature?
Write chemical equations of the following reaction :
Propanone is treated with dilute Ba (OH)2-.
Explain aldol condensation reaction in detail.
Cannizaro’s reaction is not given by ______.
Which of the following compounds do not undergo aldol condensation?
(i) \[\ce{CH3 - CHO}\]
(ii) 
(iii) \[\begin{array}{cc}
\phantom{}\ce{O}\\
\phantom{}||\\
\ce{CH3 - C - CH3}
\end{array}\]
(iv) \[\begin{array}{cc}
\phantom{}\ce{CH3}\\
|\phantom{...}\\
\ce{CH3 - C - CHO}\phantom{..}\\
|\phantom{...}\\
\phantom{}\ce{CH3}\\
\end{array}\]
Cross aldol condensation occurs between
Which of the following gives aldol con~ensation reaction?
Which of the following compounds will undergo self-condensation in the presence of dilute NaOH solution?
Why is the α-hydrogens of aldehydes and ketones are acidic in nature?
Assertion (A): The final product in Aldol condensation is always α, β-unsaturated carbonyl compound.
Reason (R): α, β-unsaturated carbonyl compounds are stabilised due to conjugation.
What is aldol condensation? Explain it with suitable examples.
Which one of the following undergoes reaction with 50% sodiumhydroxide solution to give the corresponding alcohol and acid:
