Advertisements
Advertisements
प्रश्न
A steam engine delivers 5.4×108 J of work per minute and services 3.6 × 109 J of heat per minute from its boiler. What is the efficiency of the engine? How much heat is wasted per minute?
Advertisements
उत्तर १
Work done perminute, output =` 5.4 xx 10^8 J`
Heat absorbed per minute, input = `3.6 xx 10^9 J`
Efficiency, = eta = "output"/"input" = (5.4 xx 10^8)/(3.6 xx 10^9) = 0.15
`%eta = 0.15 xx 100 = 15`
Heat energy wasted /minute
= Heat energy absorbed / minute - Useful work done / minute
`= 3.6 xx 10^9 - 5.4 xx 10^8`
`= (3.6 - 0.54) xx 10^9 = 3.06 xx10^9 J`
उत्तर २
Work done by the steam engine per minute, W = 5.4 × 108 J
Heat supplied from the boiler, H = 3.6 × 109 J
Efficiency of the engine = `"Output energy"/"Inut energy"`
`:.eta = W/H = (5.4 xx 10^8)/(3.6 xx 10^9) = 0.15`
Hence, the percentage efficiency of the engine is 15 %.
Amount of heat wasted = 3.6 × 109 – 5.4 × 108
= 30.6 × 108 = 3.06 × 109 J
Therefore, the amount of heat wasted per minute is 3.06 × 109 J.
संबंधित प्रश्न
A cylinder containing a gas is lifted from the first floor to the second floor. What is the amount of work done on the gas? What is the amount of work done by the gas? Is the internal energy of the gas increased? Is the temperature of the gas increased?
A closed bottle contains some liquid. the bottle is shaken vigorously for 5 minutes. It is found that the temperature of the liquid is increased. Is heat transferred to the liquid? Is work done on the liquid? Neglect expansion on heating.
The final volume of a system is equal to the initial volume in a certain process. Is the work done by the system necessarily zero? Is it necessarily nonzero?
An ideal gas is pumped into a rigid container having diathermic walls so that the temperature remains constant. In a certain time interval, the pressure in the container is doubled. Is the internal energy of the contents of the container also doubled in the interval ?
When a tyre bursts, the air coming out is cooler than the surrounding air. Explain.
In a process on a system, the initial pressure and volume are equal to the final pressure and volume.
(a) The initial temperature must be equal to the final temperature.
(b) The initial internal energy must be equal to the final internal energy.
(c) The net heat given to the system in the process must be zero.
(d) The net work done by the system in the process must be zero.
Which of the following system freely allows the exchange of energy and matter with its environment?
What is the internal energy of the system, when the amount of heat Q is added to the system and the system does not do any work during the process?
Explain the different ways through which the internal energy of the system can be changed.
A cylinder containing one gram molecule of the gas was compressed adiabatically until its temperature rose from 27°C to 97°C. Calculate the work done and heat produced in the gas (𝛾 = 1.5).
A thermodynamic system goes from states (i) P, V to 2P, V (ii) P, V to P, 2V. The work done in the two cases is ______.
Which of the following represents isothermal process?
In insulated systems, the amount of external work done by the gas is proportional to:
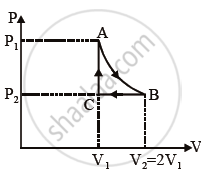
n mole of a perfect gas undergoes a cyclic process ABCA (see figure) consisting of the following processes:
A `→` B: Isothermal expansion at temperature T so that the volume is doubled from V1 to V2 = 2V1 and pressure changes from P1 to P2.
B `→` C: Isobaric compression at pressure P2 to initial volume V1.
C `→` A: Isochoric change leading to change of pressure from P2 to P1.
Total workdone in the complete cycle ABCA is ______.
An expansion process on a diatomic ideal gas (Cv = 5/2 R), has a linear path between the initial and final coordinates on a pV diagram. The coordinates of the initial state are: the pressure is 300 kPa, the volume is 0.08 m3 and the temperature is 390 K. The final pressure is 90 kPa and the final temperature s 320 K. The change in the internal energy of the gas, in SI units, is closest to:
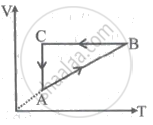
A cyclic process ABCA is shown in the V-T diagram. A process on the P-V diagram is ______.
The molar specific heat of He at constant volume is 12.47 J/mol.K. Two moles of He are heated at constant pressure. So the rise in temperature is 10 K. Find the increase in internal energy of the gas.
What is heat?
