Advertisements
Advertisements
प्रश्न
A gas is taken along the path AB as shown in figure. If 70 cal of heat is extracted from the gas in the process, calculate the change in the internal energy of the system.

Advertisements
उत्तर
Given:- 70 cal of heat is extracted from the system.
Here,
∆Q = -70 cal = -(70 × 4.2) J = -294 J
From the first law of thermodynamics, we get
∆W = P ∆ V
If P is the average pressure between points A and B and ∆V is the change in volume of the system while going from point A to B, then
∆W = `-1/2`× (200 + 500) × 103 × (150 × 10−6)
∆W = `-1/2`× 700 × 150 × 10−3
∆W = - 525 × 10−1 = - 52.5 J
Here, negative sign is taken because the final volume is less than the initial volume.
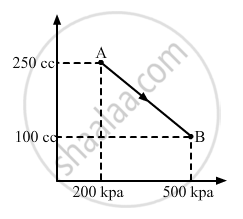
∆U = ?
∆Q = ∆U + ∆W
∆Q = - 294 J
Here, negative sign indicates that heat is extracted out from the system.
⇒ − 294 = ∆U - 52.5
⇒ ∆U = − 294 + 52.5 = - 241.5 J
APPEARS IN
संबंधित प्रश्न
A steam engine delivers 5.4×108 J of work per minute and services 3.6 × 109 J of heat per minute from its boiler. What is the efficiency of the engine? How much heat is wasted per minute?
Should the internal energy of a system necessarily increase if its temperature is increased?
A cylinder containing a gas is lifted from the first floor to the second floor. What is the amount of work done on the gas? What is the amount of work done by the gas? Is the internal energy of the gas increased? Is the temperature of the gas increased?
A force F is applied on a block of mass M. The block is displaced through a distance d in the direction of the force. What is the work done by the force on the block? Does the internal energy change because of this work?
A closed bottle contains some liquid. the bottle is shaken vigorously for 5 minutes. It is found that the temperature of the liquid is increased. Is heat transferred to the liquid? Is work done on the liquid? Neglect expansion on heating.
When a tyre bursts, the air coming out is cooler than the surrounding air. Explain.
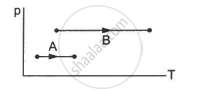
Consider two processes on a system as shown in figure.
The volumes in the initial states are the same in the two processes and the volumes in the final states are also the same. Let ∆W1 and ∆W2 be the work done by the system in the processes A and B respectively.
In a process on a system, the initial pressure and volume are equal to the final pressure and volume.
(a) The initial temperature must be equal to the final temperature.
(b) The initial internal energy must be equal to the final internal energy.
(c) The net heat given to the system in the process must be zero.
(d) The net work done by the system in the process must be zero.
A gas is initially at a pressure of 100 kPa and its volume is 2.0 m3. Its pressure is kept constant and the volume is changed from 2.0 m3 to 2.5 m3. Its Volume is now kept constant and the pressure is increased from 100 kPa to 200 kPa. The gas is brought back to its initial state, the pressure varying linearly with its volume. (a) Whether the heat is supplied to or extracted from the gas in the complete cycle? (b) How much heat was supplied or extracted?
An ideal gas is compressed at a constant temperature. Its internal energy will ____________.
In a thermodynamic system, working substance is ideal gas. Its internal energy is in the form of ______.
Two samples A and B, of a gas at the same initial temperature and pressure are compressed from volume V to V/2; A isothermally and B adiabatically. The final pressure of A will be ______.
In insulated systems, the amount of external work done by the gas is proportional to:
Figure shows the P-V diagram of an ideal gas undergoing a change of state from A to B. Four different parts I, II, III and IV as shown in the figure may lead to the same change of state.

- Change in internal energy is same in IV and III cases, but not in I and II.
- Change in internal energy is same in all the four cases.
- Work done is maximum in case I
- Work done is minimum in case II.
In thermodynamics, heat and work are ______.
An expansion process on a diatomic ideal gas (Cv = 5/2 R), has a linear path between the initial and final coordinates on a pV diagram. The coordinates of the initial state are: the pressure is 300 kPa, the volume is 0.08 m3 and the temperature is 390 K. The final pressure is 90 kPa and the final temperature s 320 K. The change in the internal energy of the gas, in SI units, is closest to:
The internal energy of one mole of argon is ______.
