Advertisements
Advertisements
प्रश्न
A substance is taken through the process abc as shown in figure. If the internal energy of the substance increases by 5000 J and a heat of 2625 cal is given to the system, calculate the value of J.

Advertisements
उत्तर
Given:-
Heat given to the system, ∆Q = 2625 cal
Increase in the internal energy of the system, ∆U = 5000 J
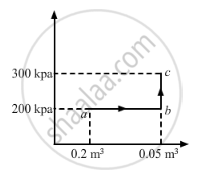
From the graph, we get
W = Area of the rectangle formed under line ab + Area under line bc
For line BC:-
Change in volume = 0
WBC = P Δ V = 0
∆W = Area of the rectangle
∆W = 200 × 103 × 0.03
= 6000 J
We know,
∆Q = ∆W + ∆U
⇒ 2625 cal = 6000 J + 5000 J
\[\Rightarrow J = \frac{11000}{2625} = 4 . 19 \text{ J/cal}\]
APPEARS IN
संबंधित प्रश्न
Explain why Air pressure in a car tyre increases during driving.
Should the internal energy of a system necessarily increase if heat is added to it?
Should the internal energy of a system necessarily increase if its temperature is increased?
A closed bottle contains some liquid. the bottle is shaken vigorously for 5 minutes. It is found that the temperature of the liquid is increased. Is heat transferred to the liquid? Is work done on the liquid? Neglect expansion on heating.
The final volume of a system is equal to the initial volume in a certain process. Is the work done by the system necessarily zero? Is it necessarily nonzero?
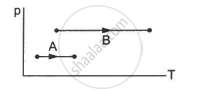
Consider two processes on a system as shown in figure.
The volumes in the initial states are the same in the two processes and the volumes in the final states are also the same. Let ∆W1 and ∆W2 be the work done by the system in the processes A and B respectively.
A mixture of fuel and oxygen is burned in a constant-volume chamber surrounded by a water bath. It was noticed that the temperature of water is increased during the process. Treating the mixture of fuel and oxygen as the system,
- Has heat been transferred?
- Has work been done?
- What is the sign of ∆U?
Which of the following is correct, when the energy is transferred to a system from its environment?
Define heat.
A system releases 100 kJ of heat while 80 kJ of work is done on the system. Calculate the change in internal energy.
One gram of water (1 cm3) becomes 1671 cm3 of steam at a pressure of 1 atm. The latent heat of vaporization at this pressure is 2256 J/g. Calculate the external work and the increase in internal energy.
The internal energy of a system is ______
Two cylinders A and B of equal capacity are connected to each other via a stopcock. A contains a gas at standard temperature and pressure. B is completely evacuated. The entire system is thermally insulated. The stopcock is suddenly opened. Answer the following:
What is the final pressure of the gas in A and B?
In insulated systems, the amount of external work done by the gas is proportional to:
A gas is compressed at a constant pressure of 50 N/m2 from a volume of 10 m3 to a volume of 4 m3. Energy of 100 J is then added to the gas by heating. Its internal energy is ______.
The internal energy of one mole of argon is ______.
A system releases 125 kJ of heat while 104 kJ work is done on the system. Calculate the change in internal energy.
Explain the change in internal energy of a thermodynamic system (the gas) by heating it.
