Advertisements
Advertisements
प्रश्न
Consider the following two statements.
(A) If heat is added to a system, its temperature must increase.
(B) If positive work is done by a system in a thermodynamic process, its volume must increase.
विकल्प
Both A and B are correct
A is correct but B is wrong
B is correct but A is wrong
Both A and B are wrong
Advertisements
उत्तर
B is correct but A is wrong
If heat is added to a system in an isothermal process, then there'll be no change in the temperature.
Work done by system, ΔW = P Δ V
⇒ ΔW = Positive ⇒ ΔV = Positive
Here,
P = Pressure
ΔV = change in volume
APPEARS IN
संबंधित प्रश्न
Explain why Air pressure in a car tyre increases during driving.
In changing the state of a gas adiabatically from an equilibrium state A to another equilibrium state B, an amount of work equal to 22.3 J is done on the system. If the gas is taken from state A to B via a process in which the net heat absorbed by the system is 9.35 cal, how much is the net work done by the system in the latter case? (Take 1 cal = 4.19 J)
The outer surface of a cylinder containing a gas is rubbed vigorously by a polishing machine. The cylinder and its gas become warm. Is the energy transferred to the gas heat or work?
A closed bottle contains some liquid. the bottle is shaken vigorously for 5 minutes. It is found that the temperature of the liquid is increased. Is heat transferred to the liquid? Is work done on the liquid? Neglect expansion on heating.
Consider two processes on a system as shown in figure.
The volumes in the initial states are the same in the two processes and the volumes in the final states are also the same. Let ∆W1 and ∆W2 be the work done by the system in the processes A and B respectively.
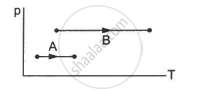
Figure shows three paths through which a gas can be taken from the state A to the state B. Calculate the work done by the gas in each of the three paths.
A substance is taken through the process abc as shown in figure. If the internal energy of the substance increases by 5000 J and a heat of 2625 cal is given to the system, calculate the value of J.

Define heat.
When does a system lose energy to its surroundings and its internal energy decreases?
Explain given cases related to energy transfer between the system and surrounding –
- energy transferred (Q) > 0
- energy transferred (Q) < 0
- energy transferred (Q) = 0
Explain the different ways through which the internal energy of the system can be changed.
The internal energy of a system is ______
A thermodynamic system goes from states (i) P, V to 2P, V (ii) P, V to P, 2V. The work done in the two cases is ______.
Figure shows the P-V diagram of an ideal gas undergoing a change of state from A to B. Four different parts I, II, III and IV as shown in the figure may lead to the same change of state.

- Change in internal energy is same in IV and III cases, but not in I and II.
- Change in internal energy is same in all the four cases.
- Work done is maximum in case I
- Work done is minimum in case II.
An expansion process on a diatomic ideal gas (Cv = 5/2 R), has a linear path between the initial and final coordinates on a pV diagram. The coordinates of the initial state are: the pressure is 300 kPa, the volume is 0.08 m3 and the temperature is 390 K. The final pressure is 90 kPa and the final temperature s 320 K. The change in the internal energy of the gas, in SI units, is closest to:
If a gas is compressed adiabatically:
A system releases 125 kJ of heat while 104 kJ work is done on the system. Calculate the change in internal energy.
