Advertisements
Advertisements
प्रश्न
Explain the different ways through which the internal energy of the system can be changed.
Advertisements
उत्तर
- The internal energy of a system can be changed by changing the temperature of the system:
a. If the system is placed in an environment that is at a temperature lower than the system, i.e., TS > TE, the energy is transferred from the system to the environment causing a decrease in the internal energy of the system.
b. If the system is placed in an environment that is at a temperature higher than the system, i.e., TE > TS, the energy is transferred from the environment to the system causing the increase in the internal energy of the system. - The internal energy of a system can be changed by doing some work:
a. When some work is done on the system by the environment, the system gains energy, and its temperature increases causing an increase in internal energy.
b. When some work is done by the system on the environment, the system loses the energy, and its temperature decreases causing a decrease in internal energy.
APPEARS IN
संबंधित प्रश्न
Explain why Two bodies at different temperatures T1 and T2, if brought in thermal contact, do not necessarily settle to the mean temperature (T1 + T2)/2.
In changing the state of a gas adiabatically from an equilibrium state A to another equilibrium state B, an amount of work equal to 22.3 J is done on the system. If the gas is taken from state A to B via a process in which the net heat absorbed by the system is 9.35 cal, how much is the net work done by the system in the latter case? (Take 1 cal = 4.19 J)
A cylinder containing a gas is lifted from the first floor to the second floor. What is the amount of work done on the gas? What is the amount of work done by the gas? Is the internal energy of the gas increased? Is the temperature of the gas increased?
The outer surface of a cylinder containing a gas is rubbed vigorously by a polishing machine. The cylinder and its gas become warm. Is the energy transferred to the gas heat or work?
A closed bottle contains some liquid. the bottle is shaken vigorously for 5 minutes. It is found that the temperature of the liquid is increased. Is heat transferred to the liquid? Is work done on the liquid? Neglect expansion on heating.
An ideal gas is pumped into a rigid container having diathermic walls so that the temperature remains constant. In a certain time interval, the pressure in the container is doubled. Is the internal energy of the contents of the container also doubled in the interval ?
Refer to figure. Let ∆U1 and ∆U2 be the changes in internal energy of the system in the process A and B. Then _____________ .
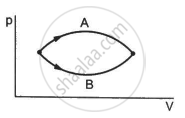
Consider the process on a system shown in figure. During the process, the work done by the system ______________ .
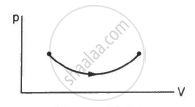
Consider two processes on a system as shown in figure.
The volumes in the initial states are the same in the two processes and the volumes in the final states are also the same. Let ∆W1 and ∆W2 be the work done by the system in the processes A and B respectively.
The pressure p and volume V of an ideal gas both increase in a process.
(a) Such a process is not possible.
(b) The work done by the system is positive.
(c) The temperature of the system must increase.
(d) Heat supplied to the gas is equal to the change in internal energy.
Figure shows a cylindrical tube of volume V with adiabatic walls containing an ideal gas. The internal energy of this ideal gas is given by 1.5 nRT. The tube is divided into two equal parts by a fixed diathermic wall. Initially, the pressure and the temperature are p1, T1 on the left and p2, T2 on the right. The system is left for sufficient time so that the temperature becomes equal on the two sides. (a) How much work has been done by the gas on the left part? (b) Find the final pressures on the two sides. (c) Find the final equilibrium temperature. (d) How much heat has flown from the gas on the right to the gas on the left?

A mixture of fuel and oxygen is burned in a constant-volume chamber surrounded by a water bath. It was noticed that the temperature of water is increased during the process. Treating the mixture of fuel and oxygen as the system,
- Has heat been transferred?
- Has work been done?
- What is the sign of ∆U?
Which of the following is correct, when the energy is transferred to a system from its environment?
Define heat.
A system releases 100 kJ of heat while 80 kJ of work is done on the system. Calculate the change in internal energy.
derive the relation between the change in internal energy (∆U), work is done (W), and heat (Q).
An ideal gas is compressed at a constant temperature. Its internal energy will ____________.
In a thermodynamic system, working substance is ideal gas. Its internal energy is in the form of ______.
8 m3 of a gas is heated at the pressure 105 N/m2 until its volume increases by 10%. Then, the external work done by the gas is ____________.
Two samples A and B, of a gas at the same initial temperature and pressure are compressed from volume V to V/2; A isothermally and B adiabatically. The final pressure of A will be ______.
Which of the following represents isothermal process?
In insulated systems, the amount of external work done by the gas is proportional to:
If a gas is compressed adiabatically:
A steam engine delivers 4.8 x 108 Jof work per minute and services 1.2 x 109 J of heat per minute from its boiler. What is the percentage efficiency of the engine?
What is heat?
A system releases 125 kJ of heat while 104 kJ work is done on the system. Calculate the change in internal energy.
Explain the change in internal energy of a thermodynamic system (the gas) by heating it.
